Evaluation of
serum level of uric acid among patients with exacerbation of asthma and
patients with controlled asthma
Azita
Tangestaninezhad1, Seyyed Ali Alavi Foumani 1, Masoud Ghasemi 1, Alireza Jafarinezhad1*
1 Inflammatory Lung Diseases Research Center, Department of Internal
Medicine, Razi Hospital, School of Medicine, Guilan University of Medical
Sciences, Rasht, Iran
*Corresponding
Author: Alireza
Jafarinezhad
* Email: alireza.jafarinezhad.md@gmail.com
Abstract
Introduction: Evidence of increased serum uric acid (UA) levels during asthma
exacerbations is still unclear. High levels of UA may lead to increased
inflammation; in this regard, we aimed to investigate the level of UA and
associated factors in patients with exacerbation of asthma attacks and those
with controlled asthma.
Materials
and Methods: In this study, demographical and clinical data from 300 patients (150
outpatients and 150 hospitalized patients) with asthma who were referred to
Razi Hospital, Rasht, Iran, from August 2018 to March 2019 were collected.
Also, the UA and spirometry parameters (FEV1, FEV1/FVC) were assessed for
patients. All data were analyzed using SPSS version 21 considering a significant
level<0.05.
Results: Among 300 patients with asthma, 158 were male, and 142 were female. A
significant association was reported between gender, body mass index (BMI),
history of smoking, opium consumption, alcohol consumption, number of asthma
attacks, family history of asthma, and history of atopy among hospitalized and
outpatients (P<0.05). In addition, there was a significant difference
between the level of UA in the two groups at the beginning of treatment
(P<0.05). Also, a significant difference between the level of UA in
hospitalized patients at the beginning and the end of treatment was observed
(P<0.05). In addition, a significant difference between the oxygen
saturation level among two groups of outpatients and hospitalized patients at
the beginning of treatment was seen (P<0.001).
Conclusion: According to our results, the level of UA might be used as a relative
predictive factor in the severity of asthma attacks.
Keywords: Asthma, Chronic obstructive pulmonary disease, Outpatients,
Hospitalized patients, Uric acid
Introduction
Asthma is a chronic
inflammatory disease of the airways characterized by increased responsiveness
of the tracheobronchial passages to various stimuli. Asthma occurs due to
temporary blockage of airflow due to chronic inflammation of the airways,
identified by periodic and reversible attacks of wheezing, shallow breathing,
shortness of breath, and cough. From the etiological point of view, asthma is a
heterogeneous disease that genetics, environment, and allergens contribute to
the onset and continuation of it (1,2). The prevalence of asthma is increasing in many
parts of the world, and it is estimated that 4-5% of the population of the USA
has asthma. Bronchial asthma occurs at any age, with the most frequent onset at
the early years of life (3).
Asthma is diagnosed according
to some symptoms, physical examination, chest X-ray, and lung function
diagnostic tests measuring FEV1, PEF, and most importantly, FEV1/FVC through
spirometry (3,4). A disproportionate response of T-helper cells
usually causes airway restriction in asthma type 2 (Th2) to allergens.
Cytokines produced by Th2 are mainly responsible for regulating many features
of asthma and lead to airway inflammation, excessive mucus secretion, and
structural changes in the airway path (5,6).
Uric acid (UA) is a product of the purine metabolism
pathway first recognized as a danger signal released from dying cells (7). It was reported that the level of UA was increased in the airways of
asthmatic patients exposed to allergens. The administration of UA crystals with
protein antigen leads to increased Th2 immunogenicity and clinical features of
asthma through dendritic cell activation, splenic tyrosine kinase, and inositol
triphosphate (IP3) kinase signaling (8).
These studies indicate that UA is an essential initiator
and enhancer of Th2 immunogenetic in asthma, reflecting airway inflammation. In
addition, strategies that target the inhibition of UA synthesis with
allopurinol or the suppression of the uricase enzyme lead to a reduction in the
production of Th2 progenitor cytokines, pulmonary inflammation, repair, and
fibrosis (9,10). Hypoxia can explain the possible potential mechanism of the impact of UA
on asthma during the exacerbation of asthma, which is induced by UA, oxidative
stress, and inflammation-inducing lung tissue damage that leads to increased
levels of UA. Also, high levels of UA may lead to increased inflammation that
ultimately impairs lung function (11,12).
Previous studies have indicated that serum levels of UA
increased in hypoxic conditions such as chronic heart failure, primary
pulmonary hypertension, and chronic obstructive pulmonary disease (COPD)
compared to hyperoxia/normoxia conditions (13–15). Moreover, the exact mechanism also occurs during the exacerbation of asthma
and bronchospasm caused by it. However, evidence of increased serum levels of
UA during asthma exacerbations is unclear; only a few studies have been
conducted on this issue. In this regard, we aimed to compare the serum level of
UA among patients with controlled asthma and patients with exacerbation of
asthma attacks at the beginning of hospitalization and at the time of discharge
from the hospital in Rasht, Iran.
Materials and Methods
Study design
This
study collected demographical data and clinical characteristics of 300 patients
(150 outpatients and 150 hospitalized patients) with asthma who were referred
to Razi hospital, Rasht, Iran, from August 2018 to March 2019. Patients with
incomplete data and a history of malignancies were excluded from the study. All
data were recorded from the patients' archives. Asthma exacerbation was diagnosed based on GINA guidelines, which include a
set of specific clinical findings, including relevant medical history,
progressive increase in shortness of breath, cough, wheezing, chest tightness,
pulse rate, respiratory rate, oxygen saturation, and peak flow measurement (16). The patients with lung diseases, in addition to asthma, suspected or
confirmed malignancy, multiple disorders or infection, acute gastrointestinal
bleeding, cardiovascular diseases, kidney failure, and consumption of foods
containing large amounts of purine, were excluded from the study. This study was
approved by the ethical committee at the Guilan University of Medical Science
[IR.GUMS.REC.1397.357].
Variables
Collected data included age,
gender, body mass index (BMI) as low weight (BMI<18.5 kg/m2),
average weight (BMI=18.5–24.99 kg/m2), overweight (BMI=25–29.9 kg/m2),
and obese (BMI≥30 kg/m2), a family history of asthma, keeping pets, history of exposure to
allergens, history of smoking, alcohol consumption, opium consumption,
underlying diseases, asthma medication, obstructive symptoms, sinusitis, mental
illness, history of reflux, atopy, level of serum UA, and spirometry indexes
such as FEV1 and FEV/FVC. In addition, the level of UV was measured by kit
(Bionik, Iran) with the BT3500 (Biotecnica Instruments. SpA -Italy)
auto-analyzer.
Statistical
analysis
All data were analyzed using
SPSS software version 21. The quantitative data were reported as mean ±
standard deviation (SD), and qualitative data were also described as numbers
and percentages. The normality was measured using the Shapiro-Wilk and
Kolmogorov-Smirnov tests. The comparison of serum levels of UA in patients with
exacerbation of asthma attacks and those with controlled asthma was measured
using the independent t-test (Mann-Whitney if were non-parametric). The
correlation between the serum level of UA and variables was measured using
Pearson's correlation test (Spearman's if were non-parametric). The comparison
of the level of UA in patients with exacerbation of asthma attack at the
beginning of hospitalization and at the time of discharge was performed using
paired t-test (Wilcoxon's if were non-parametric). Logistic regression was used
to investigate the relationship between the level of UA and asthma considering
the effects of other intervening variables. The statistical significance level
of the data was defined as P<0.05.
Results
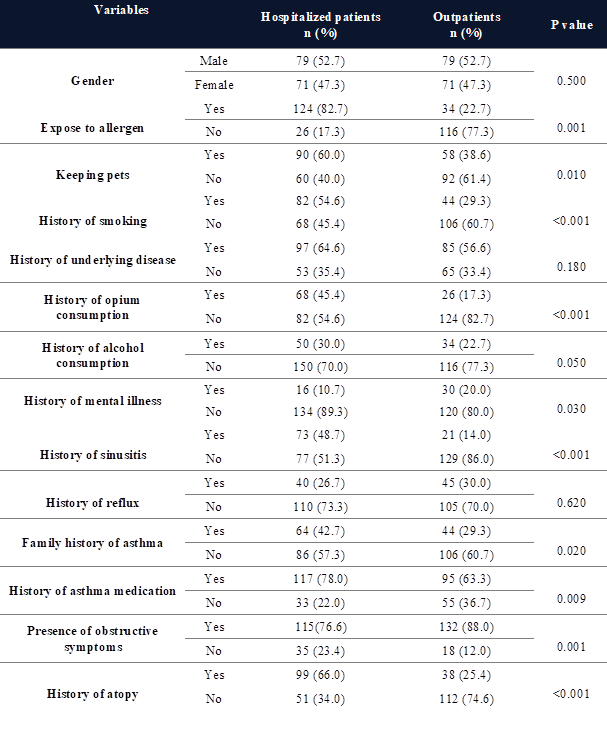
Demographical data and clinical characteristics of
patients were demonstrated in Table 1. Of the 300 studied patients, 158 were
male, and 142 were female, with a mean age of 60.19±16.48 years. The mean age
of hospitalized patients was lower than outpatients, 56.96±17.15 years (24-90)
and 60.19±15.16 years (27-86), respectively, but no statistically significant
difference was reported (P=0.610) between the mean age of two groups. The mean
BMI in hospitalized and outpatient groups was 78.78±18.98 kg/m2 and
73.96±19.22 kg/m2, respectively, representing a significant
difference between the two groups (P=0.010). The average time of asthma
diagnosis in outpatients and hospitalized groups was 88.19±117.71 months and
106.71±101.7 months, respectively (P=0.140).
Evaluating the presence or absence of daily exposure to
occupational allergens among patients revealed that most hospitalized patients
were exposed to allergens compared to the outpatients (P=0.001). Also, keeping
pets was more frequent among hospitalized patients than outpatients
(P<0.05). Most hospitalized patients had a history of opium consumption, and
the frequency of smoking was significantly higher in hospitalized patients than
in outpatients (P<0.001). There was a significant difference between the two
groups based on alcohol consumption and a history of mental illness
(P<0.05).
The average times of asthma attacks in hospitalized and
outpatients were 2.02±1.27 months (0-8), and the mean age of 66.68±21.73 years
old (1-50) during their lifetime (P<0.001). However, the frequency of
hospital administration due to asthma attacks was significantly higher in the
hospitalized group compared to the outpatients (57.12±87.78 vs. 2.09± 1.21,
P=0.010). In addition, the prevalence of a family history of asthma and
consuming asthma medication was higher among hospitalized patients (P<0.05).
Also, pulmonary construction symptoms were highly reported in both groups,
while the prevalence of atopy was significantly higher among hospitalized
patients (P<0.001).
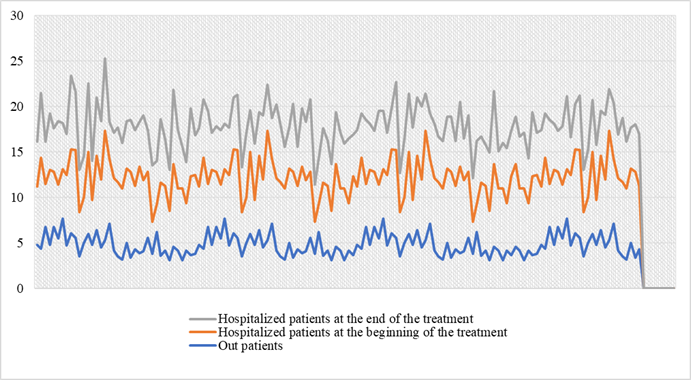
The mean serum level of UA was 4.82±1.19% mg/dL in the
outpatient group; and in the group of hospitalized patients, at the beginning
of the treatment, the mean serum level of UA was 7.31±1.85 mg/dL and at the end
of treatment was 5.69±1.11 mg/dL (P<0.001), Figure 1. There was a
significant difference between the serum level of UA in the two groups
(P<0.001). The mean oxygen saturation level in the outpatient group was
91.14±1.81%, and in hospitalized patients at the beginning and the end of
treatment, were 78.13±2.95% and 90.65±1.91%, respectively (P<0.001).
Evaluation of the level of FEV1 illustrated that in the
group of outpatients, it was 65.59 ± 3.3%. In the group of hospitalized
patients, at the beginning of the treatment, the mean serum level of UA was
69.95±44.2 mg/dL, and at the end of treatment was 94.76±62.2 mg/dL
(P<0.001). Also, the level of FVC in the group of outpatients was
65.05±2.99, and in hospitalized patients at the beginning and the end of
treatment, were 44.2±13.9 and 57.2±46.5, respectively (P<0.001).
Discussion
The present study evaluated the serum level of
UA among patients with exacerbation of asthma attacks and controlled asthma.
Most of the patients in the current study were middle-aged males (52.6%). Due
to our findings, the level of UA in hospitalized patients was significantly
higher than in outpatients. Also, the level of UA at the beginning of
hospitalization was significantly higher than at the time of discharge, similar
to other studies (17–19). In addition, our results illustrated that the level of UA was
significantly different in the two groups regarding age, gender, BMI, a history
of smoking and opium, alcohol consumption, and asthma medication (P<0.05).
This increase may be related to the asthma
attack due to the inflammatory state caused by cell necrosis and apoptosis,
followed by an increase in purine metabolites, including DNA and RNA. This increase
in metabolites elevated the function of the xanthine oxidase (XO) enzyme, which
results in higher levels of UA (20). Another mechanism that has been described as the increased UA following
an asthma attack is the increase in the degeneration of adenosine three
phosphates (ATP) and the increase in the production of UA by the XO enzyme
following hypoxia that plays a diverse role in both acute and chronic lung inflammation (21,22). In addition, several studies have mentioned the relationship between the
higher level of UA and the increase in the level of inflammatory cytokines such
as C reactive protein (CRP), tumor necrosing factor (TNF), and interleukin-1
(IL-1), which are higher in the acute phase compared to the chronic phase (11,18,23).
The current study found no significant
association between hospitalization frequency and UA level in the two groups
(P>0.05). This finding suggests that UA levels may not be a reliable marker
for predicting the likelihood of hospitalization in patients with asthma
exacerbation or controlled asthma. While previous studies have suggested a
potential link between UA and the incidence of asthma (24), our results indicate that other
factors might play a more dominant role in determining hospitalization rates.
This negative correlation implies higher UA levels may indicate poorer
respiratory function and lower oxygen saturation levels. Moreover, there was a negative association between the level of UA and FEV1 and
FVC.
These findings align with previous research suggesting that UA could contribute
to airway inflammation and obstruction, impairing lung function. While their
result of the association between hospitalization frequency
and UA level did not consistent with our study (18). However,
it is essential to note that our study only establishes an association and does
not establish a causal relationship.
A study by Lin et al. found a significant
relationship between levels of UA and functional lung disorders (17). Other studies reported that the level of UA has an inverse association
with spirometry indicators, primarily due to tissue hypoxia during the exacerbation
of asthma that causes UA production, and secondly, because the increase in UA
levels causes lung tissue inflammation and decreases lung function (25,26).
Recent studies illustrated the impact of the
antioxidants such as vitamins E and C in reducing the level of UA, which can
indicate that the level of UA may be changed due to cardiovascular diseases,
diet patterns, lifestyle, kidney function, and familial purine metabolism
problems (27,28). Moreover, the age of the patients can affect the level of UA, which
explains the variety of results in different studies (27). Also, some studies have pointed out that vitamin D, E, and C deficiency
affects the exacerbation of asthma attacks (29,30).
Limitations
One limitation of this study is referring to the recall bias of the
patients to report the duration time of asthma. Also, the impact of consuming
supplements was not investigated in the current.
Conclusions
According to our results, the level of UA at the beginning of hospitalization
was significantly higher than outpatients and at the time of discharge, which
might be used as a predictive factor for asthma severity assessment.
Author contribution
AT, SAAF, and AJ conceptualization, the original draft
writing, investigation, and formal analysis; AT and AJ conceptualization,
supervision, and project administration; SAAF and AJ conceptualization,
and project administration; AT, and MGH investigation; AJ
and MGH writing including reviewing and editing and investigation.
Conflict of interest
The authors reported no potential conflict of interest.
References
1. Price D, Fletcher M, Van Der Molen T. Asthma
control and management in 8,000 European patients: the REcognise Asthma and
LInk to Symptoms and Experience (REALISE) survey. NPJ Prim care Respir Med.
2014;24(1):1–10.
2. Hough KP, Curtiss ML,
Blain TJ, Liu R-M, Trevor J, Deshane JS, et al. Airway remodeling in asthma.
Front Med. 2020;7:191.
3. Kasper D, Fauci A,
Hauser S, Longo D, Jameson J, Loscalzo J. Harrison's principles of internal
medicine, 19e. Vol. 1. Mcgraw-hill New York, NY, USA:; 2015.
4. Burkhardt R, Pankow W.
The diagnosis of chronic obstructive pulmonary disease. Dtsch Arztebl Int. 2014
Dec;111(49):834–45, quiz 846.
5. Barnes PJ. Immunology
of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol.
2008;8(3):183–92.
6. Hammad H, Lambrecht BN.
The basic immunology of asthma. Cell. 2021;184(6):1469–85.
7. Kobayashi T, Kouzaki H,
Kita H. Human eosinophils recognize endogenous danger signal crystalline uric
acid and produce proinflammatory cytokines mediated by autocrine ATP. J
Immunol. 2010;184(11):6350–8.
8. Kool M, Willart MAM,
van Nimwegen M, Bergen I, Pouliot P, Virchow JC, et al. An unexpected role for
uric acid as an inducer of T helper 2 cell immunity to inhaled antigens and
inflammatory mediator of allergic asthma. Immunity. 2011;34(4):527–40.
9. Gasse P, Riteau N,
Charron S, Girre S, Fick L, Pétrilli V, et al. Uric acid is a danger signal
activating NALP3 inflammasome in lung injury inflammation and fibrosis. Am J
Respir Crit Care Med. 2009;179(10):903–13.
10. Lee S, Suh G-Y, Ryter
SW, Choi AMK. Regulation and function of the nucleotide binding domain
leucine-rich repeat-containing receptor, pyrin domain-containing-3 inflammasome
in lung disease. Am J Respir Cell Mol Biol. 2016;54(2):151–60.
11. Aida Y, Shibata Y, Osaka
D, Abe S, Inoue S, Fukuzaki K, et al. The relationship between serum uric acid
and spirometric values in participants in a health check: the Takahata study.
Int J Med Sci. 2011;8(6):470.
12. Hong JW, Noh JH, Kim D-J.
Association between serum uric acid and spirometric pulmonary function in
Korean adults: The 2016 Korea National Health and Nutrition Examination Survey.
PLoS One [Internet]. 2020 Oct 22;15(10):e0240987. Available from:
https://doi.org/10.1371/journal.pone.0240987
13. Rahimi-Sakak F, Maroofi
M, Rahmani J, Bellissimo N, Hekmatdoost A. Serum uric acid and risk of
cardiovascular mortality: a systematic review and dose-response meta-analysis
of cohort studies of over a million participants. BMC Cardiovasc Disord
[Internet]. 2019;19(1):218. Available from:
https://doi.org/10.1186/s12872-019-1215-z
14. Gonçalves DLN, Moreira
TR, da Silva LS. A systematic review and meta-analysis of the association
between uric acid levels and chronic kidney disease. Sci Rep. 2022;12(1):1–13.
15. Maulana S, Nuraeni A,
Aditya Nugraha B. The Potential of Prognostic Biomarkers of Uric Acid Levels in
Coronary Heart Disease Among Aged Population: A Scoping Systematic Review of
the Latest Cohort Evidence. J Multidiscip Healthc. 2022;161–73.
16. Bateman ED, Hurd SS,
Barnes PJ, Bousquet J, Drazen JM, FitzGerald M, et al. Global strategy for
asthma management and prevention: GINA executive summary. Eur Respir J.
2008;31(1):143–78.
17. Lin L, Chun W, Fuqiang
WEN. An unexpected role for serum uric acid as a biomarker for severity of
asthma exacerbation. Asian pacific J allergy Immunol. 2014;32(1):93.
18. Abdulnaby NK, Sayed AO,
Shalaby NM. Predictive value of serum uric acid in hospitalized adolescents and
adults with acute asthma. Ther Clin Risk Manag. 2016;1701–8.
19. Sayyah SG. Serum Uric
Acid Level in the Blood of Asthmatic Patients in Basrah Governorate-Iraq. J
Basrah Res. 2014;40(2).
20. Wu JT, Wu LL. Chronic
systemic inflammation leading eventually to myocardial infarction, stroke,
COPD, renal failure and cancer is induced by multiple risk factors. J Biomed
Lab Sci. 2007;19(1):1.
21. Li X, Berg NK, Mills T,
Zhang K, Eltzschig HK, Yuan X. Adenosine at the Interphase of Hypoxia and
Inflammation in Lung Injury. Front Immunol. 2020;11:604944.
22. Eltzschig HK, Carmeliet
P. Hypoxia and inflammation. N Engl J Med. 2011 Feb;364(7):656–65.
23. Zha X, Yang B, Xia G,
Wang S. Combination of uric acid and pro-inflammatory cytokines in
discriminating patients with gout from healthy controls. J Inflamm Res.
2022;1413–20.
24. Wang H, Jia Y, Yi M, Li
Y, Chen O. High Serum Uric Acid Was a Risk Factor for Incident Asthma: An Open
Cohort Study. Risk Manag Healthc Policy. 2020;13:2337–46.
25. Jeena J, Manhas S,
Prasad R, Prasad S, Gupta R. Direct Relationship Between Uric Acid and
C-Reactive Protein and Its Implication in the Chronic Kidney Disease. Indian J
Clin Biochem. 2022;37(3):365–9.
26. Ahmad A, Shameem M,
Husain Q. Relation of oxidant-antioxidant imbalance with disease progression in
patients with asthma. Ann Thorac Med. 2012;7(4):226.
27. Peruzzolo TL, Pinto JV,
Roza TH, Shintani AO, Anzolin AP, Gnielka V, et al. Inflammatory and oxidative
stress markers in post-traumatic stress disorder: a systematic review and
meta-analysis. Mol Psychiatry. 2022;1–14.
28. Al-Abdulla NO, Al Naama
LM, Hassan MK. Antioxidant status in acute asthmatic attack in children. J Pak
Med Assoc. 2010 Dec;60(12):1023–7.
29. Demirci-Çekiç S, Özkan
G, Avan AN, Uzunboy S, Çapanoğlu E, Apak R. Biomarkers of oxidative stress and
antioxidant defense. J Pharm Biomed Anal. 2022;209:114477.
30. Misso NLA,
Brooks-Wildhaber J, Ray S, Vally H, Thompson PJ. Plasma concentrations of
dietary and nondietary antioxidants are low in severe asthma. Eur Respir J.
2005;26(2):257–64.