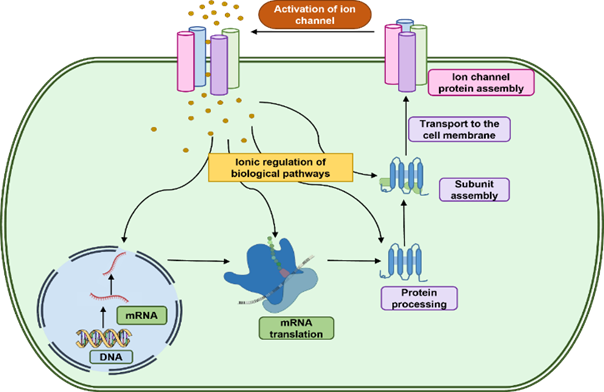
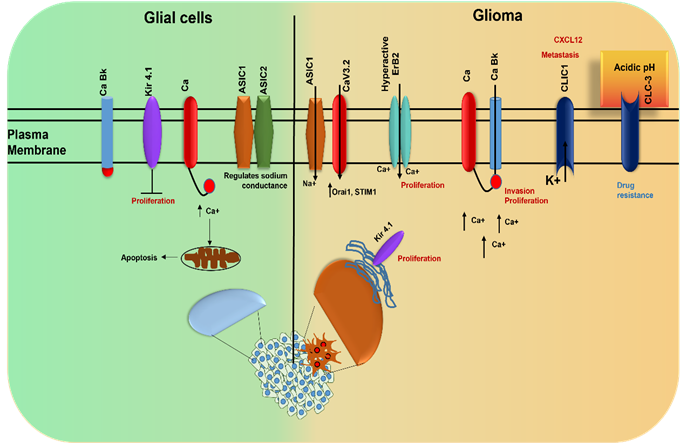
Electrophysiological analysis of cancer cells
demonstrate depolarization of the cancer cell membrane which contributes to the
stemness of cancer cells and attribute to the cellular proliferation, invasion
and migration (109, 135, 152). Studies using hepatoma and adenocarcinoma demonstrates
increased intracellular sodium concentration compared to normal cells,
suggesting a role for cancer cell depolarisation (153, 154). More recently, ion channels have been identified as
a target for cancer therapy with a few reviews summarising their role as a
therapeutic target for cancer (55,155,156). Ion channel drugs modulate ion channels by either
blocking or opening them, altering the ionic concentration and in turn, reducing
the function of oncogenic proteins across cancer cells (157,158). However, the drugs may have different modes of
action in different types of cancer due to their diverse oncogenic function and
their complex interactions between other ion channels and oncogenes (159,160). Once the altered channels are identified in any
given cancer, treating the alteration using modulators will recalibrate the
ionic balance and alter the Vm to reduce the oncogenic potential (161). Some of these drugs have been used in pre-clinical
models to demonstrate their anti-cancer effects (162–164). Based on drug repository screening in silico, a study screened over 100
United States Food and Drug Administration (FDA) approved drugs against small
cell lung cancer (SCLC) (165). Of the top 100 drugs identified, many were ion
channel modulators such as imipramine, promethazine and verapamil, that
significantly inhibited the growth of SCLC both as a single agent (165) and in combination (166). However, these drugs were specific to SCLC and did
not add any survival benefit in a phase III study in patients with
multidrug-resistant multiple myeloma (167). This suggests that ion channels may interact with
other pathways specific to the cellular context and hence, studying ion channel
drugs in the presence of other oncogenic drivers may be a greater determinant
of drug efficacy. Further, the clinical availability of multiple ion channel
drugs means a shorter translation time and a higher rate of clinical success,
underlining the need for additional research to identify roles of ion channels
in HGG tumors. However, there is limited literature exploring the opportunity
of repurposing ion channel drugs for cancer therapy (36).
Therapeutic targeting of ion channels in
HGG
HGGs are commonly treated with alkylating agents (such
as temozolomide) (168,169) and ionizing radiation (168,170) that causes DNA damage and induce apoptotic cell
death (168). However, HGG tumors develop resistance to standard
treatments due to pathway rewiring, resulting in mortality. Chemo- and
radiotherapy-induced DNA damage triggers a series of tumor-cell survival
mechanisms including alterations in calcium-activated potassium channels and
calcium-permeable non-selective cation channels (95). Ion channels are active
in excitable cells such as neurons and possess cell-specific functions (133). In vitro analyses demonstrate that activated ion channels induce
both intrinsic and acquired resistance to radiotherapy in HGG stem-like cells (95) (171). Current combination treatment with alkylating agents
and ionising agents are ineffective and additional research is required to
urgently identify alternate therapies (172). Apart from conventional drug treatments, the use of
alternative electric fields known as Tumor Treating Fields has been recently
used to treat paediatric HGG by targeting ion channels (173). Currently, this technique is being used in
combination with ion channel blockers to induce cell death, demonstrating
oncogenic role for ion channels in HGG (122). Based on the functions of ion channels in brain
cells, it is proposed that targeting aquaporin (water channels) together with
ion channels can limit HGG invasiveness, however this is yet to be tested in vivo (174).
Ion channel drugs as single-agent therapy
in HGG
CLC-3, a voltage-gated chloride channel, is present in
the plasma and intracellular membrane of HGG cells (175) and is predicted to drive drug resistance (96).
Chlorotoxin (Cltx) preferentially inhibits HGG cell invasion by binding to
CLC-3/matrix metallopeptidase 2 (MMP2) membrane complex and inhibits the
enzymatic activity of MMP2 to reduce the cell surface expression levels of MMP2
to inhibit cell invasion. Cltx has recently entered phase I and II clinical
trials for patients with HGG (176). Similarly, I-TM-601, a synthetically labelled Cltx,
has also entered phase I and II clinical trials for patients with HGG tumors (177). Besides its therapeutic potential, radiolabelled
I-TM-601 has also been used as a diagnostic marker in identifying tumor burden
in patients with HGG. Radiolabelled I-TM-601 can be effectively detected using
whole-brain single-photon emission computed tomography scans (178) and immunohistochemistry of brain tissue (179). Another study tested the efficacy of a therapeutic
antibody against CLIC1 in a pre-clinical study using HGG derived progenitor
cells where the antibody significantly increased survival in mouse models (180).
Ion channel drugs as part of combination
therapy in HGG
Multiple independent oncogenic pathways combine to
trigger the hallmarks of cancer. Combination therapy is, therefore, the gold
standard for cancer treatment (181). Ion channel drugs that failed as a single-agent
treatment have been shown to increase survival benefits when used as
combination therapy in HGG clinical trials (182). Amiodarone, an anti-arrhythmic drug, targets multiple ion channels and increases the risk of
certain types of cancers when administered for neurological disorders (183). However, in combination with a therapeutic
recombinant human protein, tumor necrosis factor-related apoptosis-inducing
ligand (TRAIL), amiodarone impeded HGG growth in vitro through the induction of calcium influx associated
apoptosis (184). This apoptotic effect was selectively in HGG cells
and did not affect normal astrocytes, highlighting the potential utility of
amiodarone as part of combination therapy for patients with HGG with minimal
side effects. This evidence together demonstrates that the pharmacodynamics of
ion channel drugs could be completely different in a cancer setting as opposed
to neurological disorders. As a result, ion channel drugs should be treated as
novel compounds for cancer treatment and stresses the importance of rigorous
pre-clinical and clinical testing. Bepridil and cibenzoline are two calcium
channel drugs that were previously used only as anti-arrhythmic agents. These
two drugs impeded HGG cell growth as part of combination therapy with TRAIL (185). The dual calcium and G-protein coupled receptor
inhibitor, pimozide, has been shown to reverse resistance to the
chemotherapeutic drug, temozolomide and sensitize HGG cells to radiation
treatment (186). Furthermore, nifedipine, a calcium channel blocker,
sensitized resistant HGG cells to cisplatin and inhibited tumor growth both in vitro and in pre-clinical mouse
models (187). In vitro
and in vivo studies demonstrate that
mibefradil, a nonspecific calcium channel blocker sensitizes HGG stem-like
cells (GSC) to temozolomide by inhibiting the AKT/mTOR pathways and
reversing stemness. Mibefradil sensitized HGG to temozolomide and stimulated
apoptotic proteins such as survivin and BAX (103). Similarly, Krouse and colleagues repurposed
mibefradil to sensitize HGG tumors to conventional chemotherapy in a phase
1b clinical trial (188). The results from the clinical trial demonstrated a
positive outcome in patients with HGG (188–190). However, despite the positive outcome in patients
with HGG, the trial could not progress further as mibefradil was withdrawn from
the market following multiple reports of drug-drug interactions (191).
Although these studies are preliminary and not all
drugs have been tested in pre-clinical animal models, the findings highlight
the potential in repurposing ion channel drugs as a treatment for patients with
HGG. However, to date, studies investigating the role of ion channels in HGG
survival have been associational, not mechanistic and further studies are
necessary to identify the mechanistic role for ion channels as a potential
therapeutic target for HGG. Pre-clinical testing of ion channel drugs on clinically
relevant HGG models will expedite the clinical translation of FDA-approved ion
channel drugs as both single and in combination with conventional therapies, in
both adult and paediatric patients with HGGs, which may otherwise cause severe
neurological side effects (192).
Research gaps in repurposing ion channel
drugs for cancer therapeutics – preclinical model development
Historically, limitations of both in vivo
animal studies and 2D in vitro models have delayed progress in this
field (193). 2D immortalized monolayer cell culture have been
used due to their cost effectiveness and reproducibility. However, they do not
accurately reflect the tumor environment, the highly heterogeneous nature of
HGG nature. Since the announcement by the United States Food and Drug
Administration in 2022 to abolish the mandate to test on animals, (through the FDA Modernization Act 2.0)
in vitro methods and models are
rapidly advancing in this area (193–195). Non-animal models include 2D cell culture, 3D
spheres (or neurospheres), organoids, bio printing, tissue-slice cultures and
tumor-on-chip methods, all which have advantages and disadvantages (194,195). To account for the highly heterogeneous nature of
HGG and for in vitro studies to be
reflective of clinical outcomes, it is important to use patient-derived cell
lines. Development of 3D in vitro models that can incorporate
vasculature and immune cell components will advance this field (194). Tumor-on-chip is both dynamic and can incorporate
vasculature by joining multiple organ (organ-on-chip) systems. Alternatively,
culturing 3D spheroids in a chip environment provides a more dynamic and
physiologically accurate model, which has already been used to test certain FDA
approved drugs for repurposing (196). More complex models have been developed since,
integrating biosensor enhanced on-chip models (197). Complex scaffolding models utilising biomaterials
have been developed to overcome some of the limitations of current models (195). Bio-banking will be essential to advance these
preclinical models. The use of preclinical models using patient-derived cell
lines can help investigate the mechanism of action and thereby, improve the
translation of research findings. Further improvements must focus on the
accurate recapitulation of the tumor microenvironment (including electrical
stimulation) and the range and plasticity of cellular states of HGG among
others (198). Combining the knowledge gleaned from computational
simulation of ion channel biology can further improve in vitro models,
particularly for tissue engineering and microfluidic (tumor-on-chip) approaches
(193,198). It is evident that ion channels play an oncogenic
role in HGG and additional research in clinically relevant models is required
for the development of effective treatments.
Future
directions
Compelling evidence on the prevalence of ion channel
aberrations in patients with HGG is starting to accumulate. However, while
ion channels are emerging as promising oncogenic targets across many different
cancers, very few ion channel inhibitors have been tested in patients with HGG (177,199).
Clinical failure is largely due to an oversight on the
interaction of ion channels with other oncogenes such as tyrosine kinases and
other ion channel family members. However, these complex interactions may be
key contributors to the cellular and pharmacological response to ion channel
drugs. For example, the same ion channel drug may induce completely different
outcomes in a patient with a kinase mutation as compared to a patient with a
transcription factor mutation. Thus, ion channels need to be studied under
the influence of other oncogenic genes in patients with HGG and
administered as personalized medicine based on the tumor pathology.
Additionally, ion channels compensate for each other’s function (52). This may explain why some drugs can effectively
impede cancer growth while others do not. Hence, it is critical to
study ionic alterations as an orchestra of multiple cellular ion channels
that maintains an electric gradient on the cell membrane to regulate cellular
processes. To achieve this, appropriate in vitro models are necessary (194,200). Testing ion channel drugs in patient
derived HGG neurospheres (201) recapitulates the clinical phenotype and hence, will
significantly increase the success of ion channel drugs in the clinic. Patient-derived
cell line mouse models will help us understand the drug’s mechanism of action
in the presence of the tumor microenvironment to bridge the knowledge gap
between the clinic and laboratory research of HGG. Personalized approaches are
essential for treating HGG due to their significant heterogeneity. These tumors
exhibit vast genetic, epigenetic, and phenotypic diversity between both
patients and within the same tumor. This variability impacts treatment
responses and outcomes, making standardized therapies less effective. By
tailoring personalized treatment strategies to the unique characteristics of
each patient's tumor, we can improve the efficacy and outcomes of patients with
HGG. With many ion channel drugs on the shelf, molecular profiling of tumor
biopsy samples to identify ion channel aberrations, can address the complex
nature of HGG more effectively with ion channel drugs.
It is important to understand the level of deviation
between the effects of ion channel drugs in
vitro and in vivo compared to the
effects they will have in clinic. Furthermore, it is necessary to undertake
studies in clinically relevant models through multi-omic approaches to better
understand the distinct oncogenic roles of ion channels in HGG. These studies
will enrich our knowledge on the molecular mechanism of ion channel drugs on
HGG tumors and help us understand drug induced resistance. These findings will
facilitate identifying a targeted therapy to treat patients with HGG with
minimal side effects.
Conclusion
The highly plastic, diffusive, and heterogeneous
nature of HGG tumors results in very low patient survival, which has been
improved only minimally with current therapeutic options due to the development
of drug resistance. The current landscape of HGG treatment is characterized by
a combination of surgery, radiotherapy, and chemotherapy, yet these approaches
often fail to extend the patient survival. Ion channels have been identified as
a promising target for HGG therapeutics, due to strong evidence of ion channel
pathologies (abnormalities/mutations) in HGG proliferation, and drug
resistance. The distinct patterns of ion channel expression observed in HGG
compared to normal brain tissues suggest that these channels play a crucial
role in HGG progression. Notably, the upregulation of certain ion channels,
such as those involved in calcium, potassium, and chloride transport,
underscores their potential as biomarkers for HGG prognosis and as targets for
therapeutic intervention. The same ion channels identified as therapeutic
targets could also be used as biomarkers for disease or disease progression.
Ion channel drugs have been repurposed for a host of neurological conditions,
and although these drugs cross the blood-brain barrier, only a few have been
tested for their anti-cancer effects. In addition, their complex mode of action
is not well understood. There is growing evidence of the potential to use ion
channel drugs for cancer therapy, particularly in combination with conventional
therapy, but differences in the pharmacodynamics of ion channel drugs need
further mechanistic investigation. The integration of ion channel-targeting
drugs into clinical protocols offers a promising avenue to enhance therapeutic
efficacy. Preclinical studies have already demonstrated the potential of ion
channel modulators in reducing HGG growth and sensitizing tumors to
conventional treatments. For instance, drugs targeting specific potassium and
chloride channels have shown encouraging results in pre-clinical models by
impairing HGG growth and inducing apoptosis.
Future research should focus on elucidating the
precise roles of various ion channels in HGG pathophysiology and identifying
ion channel-targeting compounds for clinical use. Personalized medicine
approaches, leveraging the unique ion channel expression profiles of individual
tumors, could further refine treatment strategies, ensuring maximal therapeutic
benefit while minimizing adverse effects. New 3D in vitro models using patient-derived cells, tissue engineering and
microfluidic approaches are improving the accurate recapitulation of the tumor
environment and the range and plasticity of cellular states of HGG, showing
promise to elucidate oncogenic mechanisms of HGG. Targeting ion channels
represents a novel and promising therapeutic strategy in the fight against HGG,
with the potential to significantly improve patient outcomes and advance the
current standard of care. Clinically relevant and physiologically accurate
models will improve testing of ion channel drugs, offering a personalized
medicine approach that can be combined with multi-omics to improve our
understanding of HGG, HGG drug resistance and create enhanced therapeutics with
minimal side effects.
Acknowledgment
We
thank the Pirate Ship Foundation, The Cure starts Now, Perth Children’s hospital
Foundation, The Brain Tumor Charity, and the Robert Connor Dawes Foundation for
supporting the research work related to the review. A special thanks to senior
postdoctoral researchers within the institute who mentored us in completing
this review.
Author
contribution
PJM conceptualizing, drafting, editing and
reference collection. NA
conceptualizing and overall editing, CR
drafting and editing.
Conflict
of interest
The
authors report no conflict of interest.
Funding
There
is no funding agency involved in this research.
References
1. Jäkel S,
Dimou L. Glial Cells and Their Function in the Adult Brain: A Journey through
the History of Their Ablation. Front Cell Neurosci. 2017;11:24–24.
2. Zong H,
Verhaak RGW, Canoll P. The cellular origin for malignant glioma and prospects
for clinical advancements. Expert Rev Mol Diagn. 2012 May;12(4):383–94.
3. Forsyth
PA, Wong H, Laing TD, Rewcastle NB, Morris DG, Muzik H, et al. Gelatinase-A
(MMP-2), gelatinase-B (MMP-9) and membrane type matrix metalloproteinase-1
(MT1-MMP) are involved in different aspects of the pathophysiology of malignant
gliomas. Br J Cancer. 1999 Apr;79(11–12):1828–35.
4. Turner KL,
Sontheimer H. Cl- and K+ channels and their role in primary brain tumour
biology. Philos Trans R Soc Lond B Biol Sci. 2014
Mar;369(1638):20130095–20130095.
5. Louis DN,
Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et
al. The 2016 World Health Organization Classification of Tumors of the Central
Nervous System: a summary. Acta Neuropathol (Berl). 2016 Jun;131(6):803–20.
6. Valk PE,
Dillon WP. Radiation injury of the brain. AJNR Am J Neuroradiol. 1991
Jan;12(1):45–62.
7. Stupp R,
Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJB, Janzer RC, et al. Effects of
radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy
alone on survival in glioblastoma in a randomised phase III study: 5-year
analysis of the EORTC-NCIC trial. Lancet Oncol. 2009 May;10(5):459–66.
8. Pillay V,
Allaf L, Wilding AL, Donoghue JF, Court NW, Greenall SA, et al. The plasticity
of oncogene addiction: implications for targeted therapies directed to receptor
tyrosine kinases. Neoplasia N Y N. 2009 May;11(5):448–58, 2 p following 458.
9. Mateos-Aparicio
P, Rodríguez-Moreno A. The Impact of Studying Brain Plasticity. Front Cell
Neurosci. 2019;13:66–66.
10. Abou-Antoun
TJ, Hale JS, Lathia JD, Dombrowski SM. Brain Cancer Stem Cells in Adults and
Children: Cell Biology and Therapeutic Implications. Neurother J Am Soc Exp
Neurother. 2017 Apr;14(2):372–84.
11. Simon OJ,
Müntefering T, Grauer OM, Meuth SG. The role of ion channels in malignant brain
tumors. J Neurooncol. 2015 Nov;125(2):225–35.
12. Molenaar
RJ. Ion channels in glioblastoma. ISRN Neurol. 2011;2011:590249–590249.
13. Griffin M,
Khan R, Basu S, Smith S. Ion Channels as Therapeutic Targets in High Grade
Gliomas. Cancers. 2020 Oct;12(10).
14. Kirmiz M,
Palacio S, Thapa P, King AN, Sack JT, Trimmer JS. Remodeling neuronal ER-PM
junctions is a conserved nonconducting function of Kv2 plasma membrane ion
channels. Mol Biol Cell. 2018 Oct;29(20):2410–32.
15. Chittajallu
R, Chen Y, Wang H, Yuan X, Ghiani CA, Heckman T, et al. Regulation of Kv1
subunit expression in oligodendrocyte progenitor cells and their role in G1/S
phase progression of the cell cycle. Proc Natl Acad Sci U S A. 2002
Feb;99(4):2350–5.
16. Ghiani CA,
Yuan X, Eisen AM, Knutson PL, DePinho RA, McBain CJ, et al. Voltage-activated
K+ channels and membrane depolarization regulate accumulation of the
cyclin-dependent kinase inhibitors p27(Kip1) and p21(CIP1) in glial progenitor
cells. J Neurosci Off J Soc Neurosci. 1999 Jul;19(13):5380–92.
17. Becchetti
A. Ion channels and transporters in cancer. 1. Ion channels and cell
proliferation in cancer. Am J Physiol Cell Physiol. 2011 Aug;301(2):C255-65.
18. Bortner CD,
Hughes FM, Cidlowski JA. A primary role for K+ and Na+ efflux in the activation
of apoptosis. J Biol Chem. 1997 Dec;272(51):32436–42.
19. Daoudal G,
Debanne D. Long-term plasticity of intrinsic excitability: learning rules and
mechanisms. Learn Mem Cold Spring Harb N. 2003;10(6):456–65.
20. Naoki H,
Sakumura Y, Ishii S. Local signaling with molecular diffusion as a decoder of
Ca2+ signals in synaptic plasticity. Mol Syst Biol. 2005;1:2005.0027-2005.0027.
21. Dityatev A,
Rusakov DA. Molecular signals of plasticity at the tetrapartite synapse. Curr
Opin Neurobiol. 2011 Apr;21(2):353–9.
22. Zhao X, Han
C, Zeng Z, Liu L, Wang H, Xu J, et al. Glutamate Attenuates the Survival
Property of IGFR through NR2B Containing N-Methyl-D-aspartate Receptors in
Cortical Neurons. Oxid Med Cell Longev. 2020;2020:5173184–5173184.
23. Ryland KE,
Hawkins AG, Weisenberger DJ, Punj V, Borinstein SC, Laird PW, et al. Promoter
Methylation Analysis Reveals That KCNA5 Ion Channel Silencing Supports Ewing
Sarcoma Cell Proliferation. Mol Cancer Res MCR. 2016 Jan;14(1):26–34.
24. Liu ZB, Liu
C, Zeng B, Huang LP, Yao LH. Modulation Effects of Cordycepin on Voltage-Gated
Sodium Channels in Rat Hippocampal CA1 Pyramidal Neurons in the
Presence/Absence of Oxygen. Neural Plast. 2017;2017:2459053–2459053.
25. HODGKIN AL,
HUXLEY AF. A quantitative description of membrane current and its application
to conduction and excitation in nerve. J Physiol. 1952 Aug;117(4):500–44.
26. Catacuzzeno
L, Sforna L, Esposito V, Limatola C, Franciolini F. Ion Channels in Glioma
Malignancy. Rev Physiol Biochem Pharmacol. 2021;181:223–67.
27. Liu J, Qu
C, Han C, Chen MM, An LJ, Zou W. Potassium channels and their role in glioma: A
mini review. Mol Membr Biol. 2019 Dec;35(1):76–85.
28. Varricchio
A, Ramesh SA, Yool AJ. Novel Ion Channel Targets and Drug Delivery Tools for
Controlling Glioblastoma Cell Invasiveness. Int J Mol Sci. 2021 Nov;22(21).
29. Parsons DW,
Jones S, Zhang X, Lin JCH, Leary RJ, Angenendt P, et al. An integrated genomic
analysis of human glioblastoma multiforme. Science. 2008 Sep;321(5897):1807–12.
30. Joshi AD,
Parsons DW, Velculescu VE, Riggins GJ. Sodium ion channel mutations in
glioblastoma patients correlate with shorter survival. Mol Cancer. 2011
Feb;10:17–17.
31. Sheykhzadeh
S, Luo M, Peng B, White J, Abdalla Y, Tang T, et al. Transferrin-targeted
porous silicon nanoparticles reduce glioblastoma cell migration across tight
extracellular space. Sci Rep. 2020 Feb;10(1):2320–2320.
32. Olsen ML,
Weaver AK, Ritch PS, Sontheimer H. Modulation of glioma BK channels via erbB2.
J Neurosci Res. 2005 Jul;81(2):179–89.
33. Abbott NJ,
Khan EU, Rollinson CMS, Reichel A, Janigro D, Dombrowski SM, et al. Drug
resistance in epilepsy: the role of the blood-brain barrier. Novartis Found
Symp. 2002;243:38–5.
34. Elias AF,
Lin BC, Piggott BJ. Ion Channels in Gliomas-From Molecular Basis to Treatment.
Int J Mol Sci. 2023 Jan;24(3).
35. Keluth S,
Jitte S, Bhushan R, Ranjan OP, Murti K, Ravichandiran V, et al. Targeting Ion
Channels for the Treatment of Glioma. Mini-Rev Med Chem. 2023
Jul;23(12):1298–318.
36. Kirtonia A,
Gala K, Fernandes SG, Pandya G, Pandey AK, Sethi G, et al. Repurposing of
drugs: An attractive pharmacological strategy for cancer therapeutics. Semin
Cancer Biol. 2021 Jan;68:258–78.
37. Gadsby DC.
Ion channels versus ion pumps: the principal difference, in principle. Nat Rev
Mol Cell Biol. 2009 May;10(5):344–52.
38. Anderson
KJ, Cormier RT, Scott PM. Role of ion channels in gastrointestinal cancer.
World J Gastroenterol. 2019 Oct;25(38):5732–72.
39. Green WN.
Ion channel assembly: creating structures that function. J Gen Physiol. 1999
Feb;113(2):163–70.
40. Unwin N.
The structure of ion channels in membranes of excitable cells. Neuron. 1989
Dec;3(6):665–76.
41. Isacoff EY,
Jan LY, Minor DL. Conduits of life’s spark: a perspective on ion channel
research since the birth of neuron. Neuron. 2013 Oct;80(3):658–74.
42. Kulbacka J,
Choromańska A, Rossowska J, Weżgowiec J, Saczko J, Rols MP. Cell Membrane
Transport Mechanisms: Ion Channels and Electrical Properties of Cell Membranes.
Adv Anat Embryol Cell Biol. 2017;227:39–58.
43. Fohlmeister
JF. Voltage gating by molecular subunits of Na+ and K+ ion channels:
higher-dimensional cubic kinetics, rate constants, and temperature. J
Neurophysiol. 2015 Jun;113(10):3759–77.
44. Abdul Kadir
L, Stacey M, Barrett-Jolley R. Emerging Roles of the Membrane Potential: Action
Beyond the Action Potential. Front Physiol. 2018;9:1661–1661.
45. Shah S, Chu
Y, Cegielski V, Chu XP. Acid-Sensing Ion Channel 1 Contributes to Weak
Acid-Induced Migration of Human Malignant Glioma Cells. Front Physiol.
2021;12:734418–734418.
46. Armstrong
CM. Na channel inactivation from open and closed states. Proc Natl Acad Sci.
2006 Nov;103(47):17991–6.
47. Waszkielewicz
AM, Gunia A, Szkaradek N, Słoczyńska K, Krupińska S, Marona H. Ion channels as
drug targets in central nervous system disorders. Curr Med Chem.
2013;20(10):1241–85.
48. Nelson MT,
Todorovic SM, Perez-Reyes E. The role of T-type calcium channels in epilepsy
and pain. Curr Pharm Des. 2006;12(18):2189–97.
49. Smart TG,
Paoletti P. Synaptic neurotransmitter-gated receptors. Cold Spring Harb
Perspect Biol. 2012 Mar;4(3).
50. Cheng KT,
Ong HL, Liu X, Ambudkar IS. Contribution and regulation of TRPC channels in
store-operated Ca2+ entry. Curr Top Membr. 2013;71:149–79.
51. Catterall
WA. Voltage-gated calcium channels. Cold Spring Harb Perspect Biol. 2011
Aug;3(8):a003947–a003947.
52. Rosati B,
McKinnon D. Regulation of ion channel expression. Circ Res. 2004
Apr;94(7):874–83.
53. Soriani O,
Rapetti-Mauss R. Sigma 1 Receptor and Ion Channel Dynamics in Cancer. Adv Exp
Med Biol. 2017;964:63–77.
54. Haworth AS,
Brackenbury WJ. Emerging roles for multifunctional ion channel auxiliary
subunits in cancer. Cell Calcium. 2019 Jun;80:125–40.
55. Capatina
AL, Lagos D, Brackenbury WJ. Targeting Ion Channels for Cancer Treatment:
Current Progress and Future Challenges. Rev Physiol Biochem Pharmacol.
2022;183:1–43.
56. Litan A,
Langhans SA. Cancer as a channelopathy: ion channels and pumps in tumor
development and progression. Front Cell Neurosci. 2015;9:86–86.
57. Pedersen
SF, Stock C. Ion channels and transporters in cancer: pathophysiology,
regulation, and clinical potential. Cancer Res. 2013 Mar;73(6):1658–61.
58. Farfariello
V, Prevarskaya N, Gkika D. Ion Channel Profiling in Prostate Cancer: Toward
Cell Population-Specific Screening. Rev Physiol Biochem Pharmacol.
2021;181:39–56.
59. Prevarskaya
N, Skryma R, Shuba Y. Ion Channels in Cancer: Are Cancer Hallmarks
Oncochannelopathies? Physiol Rev. 2018 Apr;98(2):559–621.
60. Shuttleworth
TJ. Intracellular Ca2+ signalling in secretory cells. J Exp Biol. 1997
Jan;200(Pt 2):303–14.
61. Rizzuto R,
De Stefani D, Raffaello A, Mammucari C. Mitochondria as sensors and regulators
of calcium signalling. Nat Rev Mol Cell Biol. 2012 Sep;13(9):566–78.
62. Kondratskyi
A, Yassine M, Kondratska K, Skryma R, Slomianny C, Prevarskaya N.
Calcium-permeable ion channels in control of autophagy and cancer. Front
Physiol. 2013;4:272–272.
63. Braun M,
Ramracheya R, Bengtsson M, Zhang Q, Karanauskaite J, Partridge C, et al.
Voltage-gated ion channels in human pancreatic beta-cells: electrophysiological
characterization and role in insulin secretion. Diabetes. 2008
Jun;57(6):1618–28.
64. Roux B. Ion
channels and ion selectivity. Essays Biochem. 2017 May;61(2):201–9.
65. Talvenheimo
JA. The purification of ion channels from excitable cells. J Membr Biol.
1985;87(2):77–91.
66. Gusovsky F,
Daly JW. Formation of second messengers in response to activation of ion
channels in excitable cells. Cell Mol Neurobiol. 1988 Jun;8(2):157–69.
67. Bertrán GC,
D’Alessio C, Kotsias BA. [Ion channels in non excitable cells]. Medicina (Mex).
1995;55(5 Pt 1):449–56.
68. Kramer RH,
Chambers JJ, Trauner D. Photochemical tools for remote control of ion channels
in excitable cells. Nat Chem Biol. 2005 Dec;1(7):360–5.
69. Deng XL,
Lau CP, Lai K, Cheung KF, Lau GK, Li GR. Cell cycle-dependent expression of
potassium channels and cell proliferation in rat mesenchymal stem cells from
bone marrow. Cell Prolif. 2007 Oct;40(5):656–70.
70. Urrego D,
Tomczak AP, Zahed F, Stühmer W, Pardo LA. Potassium channels in cell cycle and
cell proliferation. Philos Trans R Soc Lond B Biol Sci. 2014
Mar;369(1638):20130094–20130094.
71. Ekhterae D,
Platoshyn O, Krick S, Yu Y, McDaniel SS, Yuan JX. Bcl-2 decreases voltage-gated
K+ channel activity and enhances survival in vascular smooth muscle cells. Am J
Physiol Cell Physiol. 2001 Jul;281(1):C157-65.
72. Williams
SS, French JN, Gilbert M, Rangaswami AA, Walleczek J, Knox SJ. Bcl-2
overexpression results in enhanced capacitative calcium entry and resistance to
SKF-96365-induced apoptosis. Cancer Res. 2000 Aug;60(16):4358–61.
73. Roden DM,
Kupershmidt S. From genes to channels: normal mechanisms. Cardiovasc Res. 1999
May;42(2):318–26.
74. Wickenden
A, Priest B, Erdemli G. Ion channel drug discovery: challenges and future
directions. Future Med Chem. 2012 Apr;4(5):661–79.
75. Chen H,
Ikeda SR. Modulation of ion channels and synaptic transmission by a human
sensory neuron-specific G-protein-coupled receptor, SNSR4/mrgX1, heterologously
expressed in cultured rat neurons. J Neurosci Off J Soc Neurosci. 2004
May;24(21):5044–53.
76. Anderson
AJ, Harvey AL. Effects of the potassium channel blocking dendrotoxins on
acetylcholine release and motor nerve terminal activity. Br J Pharmacol. 1988
Jan;93(1):215–21.
77. Ge L, Hoa
NT, Wilson Z, Arismendi-Morillo G, Kong XT, Tajhya RB, et al. Big Potassium
(BK) ion channels in biology, disease and possible targets for cancer
immunotherapy. Int Immunopharmacol. 2014 Oct;22(2):427–43.
78. Leanza L,
Biasutto L, Managò A, Gulbins E, Zoratti M, Szabò I. Intracellular ion channels
and cancer. Front Physiol. 2013 Sep;4:227–227.
79. Peruzzo R,
Biasutto L, Szabò I, Leanza L. Impact of intracellular ion channels on cancer
development and progression. Eur Biophys J EBJ. 2016 Oct;45(7):685–707.
80. Lev S,
Moreno H, Martinez R, Canoll P, Peles E, Musacchio JM, et al. Protein tyrosine
kinase PYK2 involved in Ca(2+)-induced regulation of ion channel and MAP kinase
functions. Nature. 1995 Aug;376(6543):737–45.
81. Cheng W,
Zhu Y, Wang H. The MAPK pathway is involved in the regulation of rapid
pacing-induced ionic channel remodeling in rat atrial myocytes. Mol Med Rep.
2016 Mar;13(3):2677–82.
82. Melnikova
IN, Gardner PD. The signal transduction pathway underlying ion channel gene
regulation by SP1-C-Jun interactions. J Biol Chem. 2001 Jun;276(22):19040–5.
83. Bahcheli
AT, Min HK, Bayati M, Zhao H, Fortuna A, Dong W, et al. Pan-cancer ion
transport signature reveals functional regulators of glioblastoma aggression.
EMBO J. 2024 Jan;43(2):196–224.
84. Hoffmann
EK, Lambert IH. Ion channels and transporters in the development of drug
resistance in cancer cells. Philos Trans R Soc Lond B Biol Sci. 2014
Mar;369(1638):20130109–20130109.
85. Szado T,
Vanderheyden V, Parys JB, De Smedt H, Rietdorf K, Kotelevets L, et al.
Phosphorylation of inositol 1,4,5-trisphosphate receptors by protein kinase
B/Akt inhibits Ca2+ release and apoptosis. Proc Natl Acad Sci U S A. 2008
Feb;105(7):2427–32.
86. Liu H,
Hughes JD, Rollins S, Chen B, Perkins E. Calcium entry via ORAI1 regulates
glioblastoma cell proliferation and apoptosis. Exp Mol Pathol. 2011
Dec;91(3):753–60.
87. McFerrin
MB, Turner KL, Cuddapah VA, Sontheimer H. Differential role of IK and BK
potassium channels as mediators of intrinsic and extrinsic apoptotic cell
death. Am J Physiol Cell Physiol. 2012 Nov;303(10):C1070-8.
88. Ransom CB,
Sontheimer H. BK channels in human glioma cells. J Neurophysiol. 2001
Feb;85(2):790–803.
89. Ransom CB,
Liu X, Sontheimer H. BK channels in human glioma cells have enhanced calcium
sensitivity. Glia. 2002 Jun;38(4):281–91.
90. Liu X,
Chang Y, Reinhart PH, Sontheimer H, Chang Y. Cloning and characterization of
glioma BK, a novel BK channel isoform highly expressed in human glioma cells. J
Neurosci Off J Soc Neurosci. 2002 Mar;22(5):1840–9.
91. Olsen ML,
Schade S, Lyons SA, Amaral MD, Sontheimer H. Expression of voltage-gated
chloride channels in human glioma cells. J Neurosci Off J Soc Neurosci. 2003
Jul;23(13):5572–82.
92. Sontheimer
H. Ion channels and amino acid transporters support the growth and invasion of
primary brain tumors. Mol Neurobiol. 2004 Feb;29(1):61–71.
93. McFerrin
MB, Sontheimer H. A role for ion channels in glioma cell invasion. Neuron Glia
Biol. 2006 Feb;2(1):39–49.
94. Ruggieri P,
Mangino G, Fioretti B, Catacuzzeno L, Puca R, Ponti D, et al. The inhibition of
KCa3.1 channels activity reduces cell motility in glioblastoma derived cancer
stem cells. PloS One. 2012;7(10):e47825–e47825.
95. Huber SM,
Butz L, Stegen B, Klumpp L, Klumpp D, Eckert F. Role of ion channels in
ionizing radiation-induced cell death. Biochim Biophys Acta. 2015 Oct;1848(10
Pt B):2657–64.
96. Habela CW,
Olsen ML, Sontheimer H. ClC3 is a critical regulator of the cell cycle in
normal and malignant glial cells. J Neurosci Off J Soc Neurosci. 2008
Sep;28(37):9205–17.
97. Turner KL,
Honasoge A, Robert SM, McFerrin MM, Sontheimer H. A proinvasive role for the
Ca(2+) -activated K(+) channel KCa3.1 in malignant glioma. Glia. 2014
Jun;62(6):971–81.
98. Barbieri F,
Verduci I, Carlini V, Zona G, Pagano A, Mazzanti M, et al. Repurposed Biguanide
Drugs in Glioblastoma Exert Antiproliferative Effects via the Inhibition of
Intracellular Chloride Channel 1 Activity. Front Oncol. 2019;9:135–135.
99. García-Santisteban
I, Peters GJ, Giovannetti E, Rodríguez JA. USP1 deubiquitinase: cellular
functions, regulatory mechanisms and emerging potential as target in cancer
therapy. Mol Cancer. 2013 Aug;12:91–91.
100. Ranjan A,
Srivastava SK. Penfluridol suppresses glioblastoma tumor growth by Akt-mediated
inhibition of GLI1. Oncotarget. 2017 May;8(20):32960–76.
101. Pollak J,
Rai KG, Funk CC, Arora S, Lee E, Zhu J, et al. Ion channel expression patterns
in glioblastoma stem cells with functional and therapeutic implications for
malignancy. PloS One. 2017;12(3):e0172884–e0172884.
102. Madhavan S,
Zenklusen JC, Kotliarov Y, Sahni H, Fine HA, Buetow K. Rembrandt: helping
personalized medicine become a reality through integrative translational
research. Mol Cancer Res MCR. 2009 Feb;7(2):157–67.
103. Zhang Y,
Zhang J, Jiang D, Zhang D, Qian Z, Liu C, et al. Inhibition of T-type Ca2+
channels by endostatin attenuates human glioblastoma cell proliferation and
migration. Br J Pharmacol. 2012 Jun;166(4):1247–60.
104. Zhang Y,
Cruickshanks N, Yuan F, Wang B, Pahuski M, Wulfkuhle J, et al. Targetable
T-type Calcium Channels Drive Glioblastoma. Cancer Res. 2017
Jul;77(13):3479–90.
105. Cheng YR,
Jiang BY, Chen CC. Acid-sensing ion channels: dual function proteins for
chemo-sensing and mechano-sensing. J Biomed Sci. 2018 May;25(1):46–46.
106. Sivils A,
Yang F, Wang JQ, Chu XP. Acid-Sensing Ion Channel 2: Function and Modulation.
Membranes. 2022 Jan;12(2).
107. Berdiev BK,
Xia J, McLean LA, Markert JM, Gillespie GY, Mapstone TB, et al. Acid-sensing
ion channels in malignant gliomas. J Biol Chem. 2003 Apr;278(17):15023–34.
108. Tian Y,
Bresenitz P, Reska A, El Moussaoui L, Beier CP, Gründer S. Glioblastoma cancer
stem cell lines express functional acid sensing ion channels ASIC1a and ASIC3.
Sci Rep. 2017 Oct;7(1):13674–13674.
109. Caldwell JH,
Schaller KL, Lasher RS, Peles E, Levinson SR. Sodium channel Nav1. 6 is
localized at nodes of Ranvier, dendrites, and synapses. Proc Natl Acad Sci.
2000;97(10):5616–20.
110. Ai Y, Zhang
X, Hu X, Gao J, Liu J, Ou S, et al. Role of the voltage‑gated sodium channel
Nav1.6 in glioma and candidate drugs screening. Int J Mol Med. 2023 Jun;51(6).
111. Boyle Y,
Johns TG, Fletcher EV. Potassium Ion Channels in Malignant Central Nervous
System Cancers. Cancers. 2022 Sep;14(19).
112. Berkefeld H,
Sailer CA, Bildl W, Rohde V, Thumfart JO, Eble S, et al. BKCa-Cav channel
complexes mediate rapid and localized Ca2+-activated K+ signaling. Science.
2006 Oct;314(5799):615–20.
113. Comes N,
Bielanska J, Vallejo-Gracia A, Serrano-Albarrás A, Marruecos L, Gómez D, et al.
The voltage-dependent K+ channels Kv1. 3 and Kv1. 5 in human cancer. Front
Physiol. 2013;4:283.
114. Bielanska J,
Hernandez-Losa J, Perez-Verdaguer M, Moline T, Somoza R, Cajal S, et al.
Voltage-dependent potassium channels Kv1. 3 and Kv1. 5 in human cancer. Curr
Cancer Drug Targets. 2009;9(8):904–14.
115. Arvind S,
Arivazhagan A, Santosh V, Chandramouli BA. Differential expression of a novel
voltage gated potassium channel--Kv 1.5 in astrocytomas and its impact on
prognosis in glioblastoma. Br J Neurosurg. 2012 Feb;26(1):16–20.
116. Meizeng L,
Tian P, Qing Zhao, Xialin Ma, Yunxiang Zhang, Li. Potassium channels: Novel
targets for tumor diagnosis and chemoresistance. Front Oncol.
2023;(12):1074469.
117. Ohno Y,
Kunisawa N, Shimizu S. Emerging roles of astrocyte Kir4. 1 channels in the
pathogenesis and treatment of brain diseases. Int J Mol Sci. 2021;22(19):10236.
118. Higashimori
H, Sontheimer H. Role of Kir4.1 channels in growth control of glia. Glia. 2007
Dec;55(16):1668–79.
119. Franciolini
F, Petris A. Chloride channels of biological membranes. Biochim Biophys Acta
BBA-Rev Biomembr. 1990;1031(2):247–59.
120. Nilius B,
Droogmans G. Amazing chloride channels: an overview. Acta Physiol Scand.
2003;177(2):119–47.
121. Wang CY, Lai
MD, Phan NN, Sun Z, Lin YC. Meta-Analysis of Public Microarray Datasets Reveals
Voltage-Gated Calcium Gene Signatures in Clinical Cancer Patients. PloS One.
2015;10(7):e0125766–e0125766.
122. Griffin M,
Basu S, Khan R, Smith S. CLIC1 and CLIC4 Ion Channel Deficiency Confers
Increased Sensitivity to Tumour Treating Fields and Improved Survival in
Paediatric Glioblastoma. Neuro-Oncol. 2022 Oct;24(Supplement_4):iv4–iv4.
123. Sciaccaluga
M, Fioretti B, Catacuzzeno L, Pagani F, Bertollini C, Rosito M, et al.
CXCL12-induced glioblastoma cell migration requires intermediate conductance
Ca2+-activated K+ channel activity. Am J Physiol Cell Physiol. 2010
Jul;299(1):C175-84.
124. Cuddapah VA,
Sontheimer H. Molecular interaction and functional regulation of ClC-3 by
Ca2+/calmodulin-dependent protein kinase II (CaMKII) in human malignant glioma.
J Biol Chem. 2010;285(15):11188–96.
125. Sun P, Mu Y,
Zhang S. A novel NF-κB/MMP-3 signal pathway involves in the aggressivity of
glioma promoted by Bmi-1. Tumor Biol. 2014;35:12721–7.
126. Wang B, Xie
J, He HY, Huang EW, Cao QH, Luo L, et al. Suppression of CLC-3 chloride channel
reduces the aggressiveness of glioma through inhibiting nuclear factor-κB
pathway. Oncotarget. 2017;8(38):63788.
127. Sontheimer
H. An unexpected role for ion channels in brain tumor metastasis. Exp Biol Med
Maywood NJ. 2008 Jul;233(7):779–91.
128. Ernest NJ,
Weaver AK, Van Duyn LB, Sontheimer HW. Relative contribution of chloride
channels and transporters to regulatory volume decrease in human glioma cells.
Am J Physiol-Cell Physiol. 2005;288(6):C1451–60.
129. Chinigò G,
Castel H, Chever O, Gkika D. TRP Channels in Brain Tumors. Front Cell Dev Biol.
2021;9:617801–617801.
130. Alptekin M,
Eroglu S, Tutar E, Sencan S, Geyik MA, Ulasli M, et al. Gene expressions of TRP
channels in glioblastoma multiforme and relation with survival. Tumour Biol J
Int Soc Oncodevelopmental Biol Med. 2015 Dec;36(12):9209–13.
131. Gehring MP,
Kipper F, Nicoletti NF, Sperotto ND, Zanin R, Tamajusuku ASK, et al. P2X7
receptor as predictor gene for glioma radiosensitivity and median survival. Int
J Biochem Cell Biol. 2015 Nov;68:92–100.
132. Chen X,
Momin A, Wanggou S, Wang X, Min HK, Dou W, et al. Mechanosensitive brain tumor
cells construct blood-tumor barrier to mask chemosensitivity. Neuron. 2023
Jan;111(1):30-48.e14.
133. Qu S, Hu T,
Qiu O, Su Y, Gu J, Xia Z. Effect of Piezo1 Overexpression on Peritumoral Brain
Edema in Glioblastomas. AJNR Am J Neuroradiol. 2020 Aug;41(8):1423–9.
134. Goldman DE.
Potential, impedance, and rectification in membranes. J Gen Physiol.
1943;27(1):37–60.
135. Levin M.
Large-scale biophysics: ion flows and regeneration. Trends Cell Biol.
2007;17(6):261–70.
136. Hodgkin AL,
Katz B. The effect of sodium ions on the electrical activity of the giant axon
of the squid. J Physiol. 1949;108(1):37.
137. Imbrici P,
Liantonio A, Camerino GM, De Bellis M, Camerino C, Mele A, et al. Therapeutic
Approaches to Genetic Ion Channelopathies and Perspectives in Drug Discovery.
Front Pharmacol. 2016;7:121–121.
138. Markman JD,
Dworkin RH. Ion channel targets and treatment efficacy in neuropathic pain. J
Pain. 2006 Jan;7(1 Suppl 1):S38-47.
139. Grigoriadis
DE, Hoare SRJ, Lechner SM, Slee DH, Williams JA. Drugability of extracellular
targets: discovery of small molecule drugs targeting allosteric, functional,
and subunit-selective sites on GPCRs and ion channels. Neuropsychopharmacol Off
Publ Am Coll Neuropsychopharmacol. 2009 Jan;34(1):106–25.
140. Caban A,
Pisarczyk K, Kopacz K, Kapuśniak A, Toumi M, Rémuzat C, et al. Filling the gap
in CNS drug development: evaluation of the role of drug repurposing. J Mark
Access Health Policy. 2017;5(1):1299833.
141. McManus OB,
Garcia ML, Weaver D, Bryant M, Titus S, Herrington JB. Ion Channel Screening,
in Assay Guidance Manual. 2012.
142. Gubič Š,
Hendrickx LA, Toplak Ž, Sterle M, Peigneur S, Tomašič T, et al. Discovery of KV
1.3 ion channel inhibitors: Medicinal chemistry approaches and challenges. Med
Res Rev. 2021 Jul;41(4):2423–73.
143. Yu H bo, Li
M, Wang W ping, Wang X liang. High throughput screening technologies for ion
channels. Acta Pharmacol Sin. 2016 Jan;37(1):34–43.
144. Kwok TCY,
Ricker N, Fraser R, Chan AW, Burns A, Stanley EF, et al. A small-molecule
screen in C. elegans yields a new calcium channel antagonist. Nature. 2006
May;441(7089):91–5.
145. Burns AR,
Wallace IM, Wildenhain J, Tyers M, Giaever G, Bader GD, et al. A predictive
model for drug bioaccumulation and bioactivity in Caenorhabditis elegans. Nat
Chem Biol. 2010 Jul;6(7):549–57.
146. Jiang Q, Li
K, Lu WJ, Li S, Chen X, Liu XJ, et al. Identification of small-molecule ion
channel modulators in C. elegans channelopathy models. Nat Commun. 2018
Sep;9(1):3941–3941.
147. Clare JJ.
Targeting ion channels for drug discovery. Discov Med. 2010 Mar;9(46):253–60.
148. Hammarstrom
AK, Gage PW. Inhibition of oxidative metabolism increases persistent sodium
current in rat CA1 hippocampal neurons. J Physiol. 1998 Aug;510 ( Pt 3)(Pt
3):735–41.
149. Issa FA,
Mock AF, Sagasti A, Papazian DM. Spinocerebellar ataxia type 13 mutation that
is associated with disease onset in infancy disrupts axonal pathfinding during
neuronal development. Dis Model Mech. 2012 Nov;5(6):921–9.
150. Cooper EC,
Jan LY. Ion channel genes and human neurological disease: recent progress,
prospects, and challenges. Proc Natl Acad Sci U S A. 1999 Apr;96(9):4759–66.
151. Lipkind GM,
Fozzard HA. Molecular model of anticonvulsant drug binding to the voltage-gated
sodium channel inner pore. Mol Pharmacol. 2010 Oct;78(4):631–8.
152. Mycielska
ME, Palmer CP, Brackenbury WJ, Djamgoz MB. Expression of Na+‐dependent citrate
transport in a strongly metastatic human prostate cancer PC‐3M cell line:
regulation by voltage‐gated Na+ channel activity. J Physiol.
2005;563(2):393–408.
153. Cameron IL,
Smith NK, Pool T, Sparks R. Intracellular concentration of sodium and other
elements as related to mitogenesis and oncogenesis in vivo. Cancer Res.
1980;40(5):1493–500.
154. Yang M,
Kozminski DJ, Wold LA, Modak R, Calhoun JD, Isom LL, et al. Therapeutic
potential for phenytoin: Targeting Na v 1.5 sodium channels to reduce migration
and invasion in metastatic breast cancer. Breast Cancer Res Treat.
2012;134:603–15.
155. Szabo I,
Zoratti M, Biasutto L. Targeting mitochondrial ion channels for cancer therapy.
Redox Biol. 2021 Jun;42:101846–101846.
156. Yang J, Yu
G, Sessler JL, Shin I, Gale PA, Huang F. Artificial transmembrane ion
transporters as potential therapeutics. Chem. 2021 Dec;7(12):3256–91.
157. Prevarskaya
N, Skryma R, Shuba Y. Ion channels and the hallmarks of cancer. Trends Mol Med.
2010;16(3):107–21.
158. Arcangeli A,
Becchetti A. Integrin structure and functional relation with ion channels.
Integrins Ion Channels Mol Complexes Signal. 2010;1–7.
159. Djamgoz MB,
Mycielska M, Madeja Z, Fraser SP, Korohoda W. Directional movement of rat
prostate cancer cells in direct-current electric field: involvement of
voltagegated Na+ channel activity. J Cell Sci. 2001;114(14):2697–705.
160. K K. Ion
channels and cancer. J Membr Biol. 2005;(205):159–73.
161. Lang F,
Stournaras C. Ion channels in cancer: future perspectives and clinical
potential. Philos Trans R Soc Lond B Biol Sci. 2014
Mar;369(1638):20130108–20130108.
162. Dai X, Wang
D, Zhang J. Programmed cell death, redox imbalance, and cancer therapeutics.
Apoptosis Int J Program Cell Death. 2021 Aug;26(7–8):385–414.
163. Azimi I,
Robitaille M, Armitage K, So CL, Milevskiy MJG, Northwood K, et al. Activation
of the Ion Channel TRPV4 Induces Epithelial to Mesenchymal Transition in Breast
Cancer Cells. Int J Mol Sci. 2020 Dec;21(24).
164. Nelson M,
Yang M, Dowle AA, Thomas JR, Brackenbury WJ. The sodium channel-blocking
antiepileptic drug phenytoin inhibits breast tumour growth and metastasis. Mol
Cancer. 2015 Jan;14(1):13–13.
165. Jahchan NS,
Dudley JT, Mazur PK, Flores N, Yang D, Palmerton A, et al. A drug repositioning
approach identifies tricyclic antidepressants as inhibitors of small cell lung
cancer and other neuroendocrine tumors. Cancer Discov. 2013 Dec;3(12):1364–77.
166. Millward MJ,
Cantwell BM, Munro NC, Robinson A, Corris PA, Harris AL. Oral verapamil with
chemotherapy for advanced non-small cell lung cancer: a randomised study. Br J
Cancer. 1993 May;67(5):1031–5.
167. Dalton WS,
Crowley JJ, Salmon SS, Grogan TM, Laufman LR, Weiss GR, et al. A phase III
randomized study of oral verapamil as a chemosensitizer to reverse drug
resistance in patients with refractory myeloma. A Southwest Oncology Group
study. Cancer. 1995 Feb;75(3):815–20.
168. Sarkaria JN,
Kitange GJ, James CD, Plummer R, Calvert H, Weller M, et al. Mechanisms of
chemoresistance to alkylating agents in malignant glioma. Clin Cancer Res Off J
Am Assoc Cancer Res. 2008 May;14(10):2900–8.
169. Friedman HS,
McLendon RE, Kerby T, Dugan M, Bigner SH, Henry AJ, et al. DNA mismatch repair
and O6-alkylguanine-DNA alkyltransferase analysis and response to Temodal in
newly diagnosed malignant glioma. J Clin Oncol Off J Am Soc Clin Oncol. 1998
Dec;16(12):3851–7.
170. Simonetti G,
Gaviani P, Botturi A, Innocenti A, Lamperti E, Silvani A. Clinical management
of grade III oligodendroglioma. Cancer Manag Res. 2015;7:213–23.
171. Jamal M,
Rath BH, Tsang PS, Camphausen K, Tofilon PJ. The brain microenvironment
preferentially enhances the radioresistance of CD133(+) glioblastoma stem-like
cells. Neoplasia N Y N. 2012 Feb;14(2):150–8.
172. Blaney SM,
Poplack DG. Pharmacologic strategies for the treatment of meningeal malignancy.
Invest New Drugs. 1996;14:69–85.
173. Rominiyi O,
Vanderlinden A, Clenton SJ, Bridgewater C, Al-Tamimi Y, Collis SJ. Tumour
treating fields therapy for glioblastoma: current advances and future
directions. Br J Cancer. 2021;124(4):697–709.
174. Varricchio
A, Yool AJ. Aquaporins and Ion Channels as Dual Targets in the Design of Novel
Glioblastoma Therapeutics to Limit Invasiveness. Cancers. 2023 Jan;15(3).
175. Xu B, Mao J,
Wang L, Zhu L, Li H, Wang W, et al. ClC-3 chloride channels are essential for
cell proliferation and cell cycle progression in nasopharyngeal carcinoma
cells. Acta Biochim Biophys Sin. 2010 Jun;42(6):370–80.
176. Kasai T,
Nakamura K, Vaidyanath A, Chen L, Sekhar S, El-Ghlban S, et al. Chlorotoxin
Fused to IgG-Fc Inhibits Glioblastoma Cell Motility via Receptor-Mediated
Endocytosis. J Drug Deliv. 2012;2012:975763–975763.
177. Mamelak AN,
Jacoby DB. Targeted delivery of antitumoral therapy to glioma and other
malignancies with synthetic chlorotoxin (TM-601). Expert Opin Drug Deliv. 2007
Mar;4(2):175–86.
178. Hockaday DC,
Shen S, Fiveash J, Raubitschek A, Colcher D, Liu A, et al. Imaging glioma
extent with 131I-TM-601. J Nucl Med Off Publ Soc Nucl Med. 2005
Apr;46(4):580–6.
179. Lyons SA,
O’Neal J, Sontheimer H. Chlorotoxin, a scorpion-derived peptide, specifically
binds to gliomas and tumors of neuroectodermal origin. Glia. 2002
Aug;39(2):162–73.
180. Setti M,
Savalli N, Osti D, Richichi C, Angelini M, Brescia P, et al. Functional role of
CLIC1 ion channel in glioblastoma-derived stem/progenitor cells. J Natl Cancer
Inst. 2013 Nov;105(21):1644–55.
181. Bayat
Mokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B, et al.
Combination therapy in combating cancer. Oncotarget. 2017 Jun;8(23):38022–43.
182. Mathews J,
Kuchling F, Baez-Nieto D, Diberardinis M, Pan JQ, Levin M. Ion Channel Drugs
Suppress Cancer Phenotype in NG108-15 and U87 Cells: Toward Novel
Electroceuticals for Glioblastoma. Cancers. 2022 Mar;14(6).
183. Su VYF, Hu
YW, Chou KT, Ou SM, Lee YC, Lin EYH, et al. Amiodarone and the risk of cancer:
a nationwide population-based study. Cancer. 2013 May;119(9):1699–705.
184. Kim BJ, Kim
SY, Lee S, Jeon JH, Matsui H, Kwon YK, et al. The role of transient receptor
potential channel blockers in human gastric cancer cell viability. Can J
Physiol Pharmacol. 2012 Feb;90(2):175–86.
185. Kim IY, Kang
YJ, Yoon MJ, Kim EH, Kim SU, Kwon TK, et al. Amiodarone sensitizes human glioma
cells but not astrocytes to TRAIL-induced apoptosis via CHOP-mediated DR5
upregulation. Neuro-Oncol. 2011 Mar;13(3):267–79.
186. Rath BH,
Camphausen K, Tofilon PJ. Glioblastoma radiosensitization by pimozide. Transl
Cancer Res. 2016 Nov;5(Suppl 6):S1029–32.
187. Kondo S, Yin
D, Morimura T, Kubo H, Nakatsu S, Takeuchi J. Combination therapy with
cisplatin and nifedipine induces apoptosis in cisplatin-sensitive and
cisplatin-resistant human glioblastoma cells. Br J Cancer. 1995
Feb;71(2):282–9.
188. Krouse AJ,
Gray L, Macdonald T, McCray J. Repurposing and Rescuing of Mibefradil, an
Antihypertensive, for Cancer: A Case Study. Assay Drug Dev Technol. 2015
Dec;13(10):650–3.
189. Holdhoff M,
Ye X, Supko JG, Nabors LB, Desai AS, Walbert T, et al. Timed sequential therapy
of the selective T-type calcium channel blocker mibefradil and temozolomide in
patients with recurrent high-grade gliomas. Neuro-Oncol. 2017 Jun;19(6):845–52.
190. Keir ST,
Friedman HS, Reardon DA, Bigner DD, Gray LA. Mibefradil, a novel therapy for
glioblastoma multiforme: cell cycle synchronization and interlaced therapy in a
murine model. J Neurooncol. 2013 Jan;111(2):97–102.
191. Mullins ME,
Horowitz BZ, Linden DH, Smith GW, Norton RL, Stump J. Life-threatening
interaction of mibefradil and beta-blockers with dihydropyridine calcium
channel blockers. JAMA. 1998 Jul;280(2):157–8.
192. Papin JA,
Hunter T, Palsson BO, Subramaniam S. Reconstruction of cellular signalling
networks and analysis of their properties. Nat Rev Mol Cell Biol. 2005
Feb;6(2):99–111.
193. Higginbottom
SL, Tomaskovic-Crook E, Crook JM. Considerations for modelling diffuse
high-grade gliomas and developing clinically relevant therapies. Cancer
Metastasis Rev. 2023;42(2):507–41.
194. Zottel A,
Jovčevska I, Šamec N. Non-animal glioblastoma models for personalized
treatment. Heliyon. 2023;
195. Ahmed T.
Biomaterial-based in vitro 3D modeling of glioblastoma multiforme. Cancer
Pathog Ther. 2023;1(03):177–94.
196. Fan Y,
Nguyen DT, Akay Y, Xu F, Akay M. Engineering a brain cancer chip for
high-throughput drug screening. Sci Rep. 2016;6(1):25062.
197. Thenuwara G,
Javed B, Singh B, Tian F. Biosensor-Enhanced Organ-on-a-Chip Models for
Investigating Glioblastoma Tumor Microenvironment Dynamics. Sensors.
2024;24(9):2865.
198. Maffeo C,
Bhattacharya S, Yoo J, Wells D, Aksimentiev A. Modeling and simulation of ion
channels. Chem Rev. 2012;112(12):6250–84.
199. Mamelak AN,
Rosenfeld S, Bucholz R, Raubitschek A, Nabors LB, Fiveash JB, et al. Phase I
single-dose study of intracavitary-administered iodine-131-TM-601 in adults
with recurrent high-grade glioma. J Clin Oncol Off J Am Soc Clin Oncol. 2006
Aug;24(22):3644–50.
200. FOLGIERO V,
ROMANIA P, ROSSI F, CAFORIO M, NIC M, DIBUSZ K, et al. Advanced Non-animal
Models in Biomedical Research. 2020;
201. Stringer BW,
Day BW, D’Souza RCJ, Jamieson PR, Ensbey KS, Bruce ZC, et al. A reference
collection of patient-derived cell line and xenograft models of proneural,
classical and mesenchymal glioblastoma. Sci Rep. 2019 Mar;9(1):4902–4902.
202. Chen JP,
Luan Y, You CX, Chen XH, Luo RC, Li R. TRPM7 regulates the migration of human
nasopharyngeal carcinoma cell by mediating Ca(2+) influx. Cell Calcium. 2010
May;47(5):425–32.
203. Ding X, He
Z, Shi Y, Wang Q, Wang Y. Targeting TRPC6 channels in oesophageal carcinoma
growth. Expert Opin Ther Targets. 2010 May;14(5):513–27.
204. Shiozaki A,
Kudou M, Ichikawa D, Fujiwara H, Shimizu H, Ishimoto T, et al. Esophageal
cancer stem cells are suppressed by tranilast, a TRPV2 channel inhibitor. J
Gastroenterol. 2018 Feb;53(2):197–207.
205. Choi S, Cui
C, Luo Y, Kim SH, Ko JK, Huo X, et al. Selective inhibitory effects of zinc on
cell proliferation in esophageal squamous cell carcinoma through Orai1. FASEB J
Off Publ Fed Am Soc Exp Biol. 2018 Jan;32(1):404–16.
206. Asghar MY,
Magnusson M, Kemppainen K, Sukumaran P, Löf C, Pulli I, et al. Transient
Receptor Potential Canonical 1 (TRPC1) Channels as Regulators of Sphingolipid
and VEGF Receptor Expression: IMPLICATIONS FOR THYROID CANCER CELL MIGRATION
AND PROLIFERATION. J Biol Chem. 2015 Jun;290(26):16116–31.
207. Shapovalov
G, Ritaine A, Skryma R, Prevarskaya N. Role of TRP ion channels in cancer and
tumorigenesis. Semin Immunopathol. 2016 May;38(3):357–69.
208. Wu H, Wang
H, Guan S, Zhang J, Chen Q, Wang X, et al. Cell-specific regulation of
proliferation by Ano1/TMEM16A in breast cancer with different ER, PR, and HER2
status. Oncotarget. 2017 Oct;8(49):84996–5013.
209. Elbaz M,
Ahirwar D, Xiaoli Z, Zhou X, Lustberg M, Nasser MW, et al. TRPV2 is a novel
biomarker and therapeutic target in triple negative breast cancer. Oncotarget.
2018 Sep;9(71):33459–70.
210. Badaoui M,
Mimsy-Julienne C, Saby C, Van Gulick L, Peretti M, Jeannesson P, et al.
Collagen type 1 promotes survival of human breast cancer cells by
overexpressing Kv10.1 potassium and Orai1 calcium channels through
DDR1-dependent pathway. Oncotarget. 2018 May;9(37):24653–71.
211. Potier M,
Chantome A, Joulin V, Girault A, Roger S, Besson P, et al. The SK3/K(Ca)2.3
potassium channel is a new cellular target for edelfosine. Br J Pharmacol. 2011
Jan;162(2):464–79.
212. Hall DP,
Cost NG, Hegde S, Kellner E, Mikhaylova O, Stratton Y, et al. TRPM3 and miR-204
establish a regulatory circuit that controls oncogenic autophagy in clear cell
renal cell carcinoma. Cancer Cell. 2014 Nov;26(5):738–53.
213. Song J, Wang
Y, Li X, Shen Y, Yin M, Guo Y, et al. Critical role of TRPC6 channels in the
development of human renal cell carcinoma. Mol Biol Rep. 2013
Aug;40(8):5115–22.
214. Kim JH,
Lkhagvadorj S, Lee MR, Hwang KH, Chung HC, Jung JH, et al. Orai1 and STIM1 are
critical for cell migration and proliferation of clear cell renal cell
carcinoma. Biochem Biophys Res Commun. 2014 May;448(1):76–82.
215. Rabjerg M,
Oliván-Viguera A, Hansen LK, Jensen L, Sevelsted-Møller L, Walter S, et al.
High expression of KCa3.1 in patients with clear cell renal carcinoma predicts
high metastatic risk and poor survival. PloS One. 2015;10(4):e0122992–e0122992.
216. Wang R,
Gurguis CI, Gu W, Ko EA, Lim I, Bang H, et al. Ion channel gene expression
predicts survival in glioma patients. Sci Rep. 2015 Aug;5:11593–11593.
217. Peng J, Ou
Q, Wu X, Zhang R, Zhao Q, Jiang W, et al. Expression of voltage-gated sodium
channel Nav1.5 in non-metastatic colon cancer and its associations with
estrogen receptor (ER)-β expression and clinical outcomes. Chin J Cancer. 2017
Nov;36(1):89–89.
218. Song P, Du
Y, Song W, Chen H, Xuan Z, Zhao L, et al. KCa3.1 as an Effective Target for
Inhibition of Growth and Progression of Intrahepatic Cholangiocarcinoma. J
Cancer. 2017;8(9):1568–78.
219. Ye Y, Yin M,
Huang B, Wang Y, Li X, Lou G. CLIC1 a novel biomarker of intraperitoneal
metastasis in serous epithelial ovarian cancer. Tumour Biol J Int Soc
Oncodevelopmental Biol Med. 2015 Jun;36(6):4175–9.
220. Yang SL, Cao
Q, Zhou KC, Feng YJ, Wang YZ. Transient receptor potential channel C3
contributes to the progression of human ovarian cancer. Oncogene. 2009
Mar;28(10):1320–8.
221. Gao R, Shen
Y, Cai J, Lei M, Wang Z. Expression of voltage-gated sodium channel alpha
subunit in human ovarian cancer. Oncol Rep. 2010 May;23(5):1293–9.
222. Farias LMB,
Ocaña DB, Díaz L, Larrea F, Avila-Chávez E, Cadena A, et al. Ether a go-go
potassium channels as human cervical cancer markers. Cancer Res. 2004
Oct;64(19):6996–7001.
223. Hernandez-Plata
E, Ortiz CS, Marquina-Castillo B, Medina-Martinez I, Alfaro A, Berumen J, et
al. Overexpression of NaV 1.6 channels is associated with the invasion capacity
of human cervical cancer. Int J Cancer. 2012 May;130(9):2013–23.
224. Arcangeli A,
Romoli MR, Boni L, Gerlini G, Tofani L, Urso C, et al. High hERG1 expression in
advanced melanoma. Melanoma Res. 2013 Jun;23(3):185–90.
225. Cao R, Meng
Z, Liu T, Wang G, Qian G, Cao T, et al. Decreased TRPM7 inhibits activities and
induces apoptosis of bladder cancer cells via ERK1/2 pathway. Oncotarget. 2016
Nov;7(45):72941–60.
226. Tian Y, Guan
Y, Jia Y, Meng Q, Yang J. Chloride intracellular channel 1 regulates prostate
cancer cell proliferation and migration through the MAPK/ERK pathway. Cancer
Biother Radiopharm. 2014 Oct;29(8):339–44.
227. Zhang GM,
Wan FN, Qin XJ, Cao DL, Zhang HL, Zhu Y, et al. Prognostic significance of the
TREK-1 K2P potassium channels in prostate cancer. Oncotarget. 2015
Jul;6(21):18460–8.
228. Diss JKJ,
Stewart D, Pani F, Foster CS, Walker MM, Patel A, et al. A potential novel
marker for human prostate cancer: voltage-gated sodium channel expression in
vivo. Prostate Cancer Prostatic Dis. 2005;8(3):266–73.
229. Lu J, Dong
Q, Zhang B, Wang X, Ye B, Zhang F, et al. Chloride intracellular channel 1
(CLIC1) is activated and functions as an oncogene in pancreatic cancer. Med
Oncol Northwood Lond Engl. 2015 Jun;32(6):616–616.
230. Yee NS, Chan
AS, Yee JD, Yee RK. TRPM7 and TRPM8 Ion Channels in Pancreatic Adenocarcinoma:
Potential Roles as Cancer Biomarkers and Targets. Scientifica.
2012;2012:415158–415158.
231. Yee NS,
Brown RD, Lee MS, Zhou W, Jensen C, Gerke H, et al. TRPM8 ion channel is
aberrantly expressed and required for preventing replicative senescence in
pancreatic adenocarcinoma: potential role of TRPM8 as a biomarker and target.
Cancer Biol Ther. 2012 Jun;13(8):592–9.
232. Chen CD,
Wang CS, Huang YH, Chien KY, Liang Y, Chen WJ, et al. Overexpression of CLIC1
in human gastric carcinoma and its clinicopathological significance.
Proteomics. 2007 Jan;7(1):155–67.
233. Xia J, Wang
H, Huang H, Sun L, Dong S, Huang N, et al. Elevated Orai1 and STIM1 expressions
upregulate MACC1 expression to promote tumor cell proliferation, metabolism,
migration, and invasion in human gastric cancer. Cancer Lett. 2016
Oct;381(1):31–40.
234. Xia J, Huang
N, Huang H, Sun L, Dong S, Su J, et al. Voltage-gated sodium channel Nav 1.7
promotes gastric cancer progression through MACC1-mediated upregulation of
NHE1. Int J Cancer. 2016 Dec;139(11):2553–69.
235. Ding XW,
Yang WB, Gao S, Wang W, Li Z, Hu WM, et al. Prognostic significance of hERG1
expression in gastric cancer. Dig Dis Sci. 2010 Apr;55(4):1004–10.
236. Selli C,
Erac Y, Kosova B, Erdal ES, Tosun M. Silencing of TRPC1 regulates
store-operated calcium entry and proliferation in Huh7 hepatocellular carcinoma
cells. Biomed Pharmacother Biomedecine Pharmacother. 2015 Apr;71:194–200.
237. Zhang S,
Wang XM, Yin ZY, Zhao WX, Zhou JY, Zhao BX, et al. Chloride intracellular
channel 1 is overexpression in hepatic tumor and correlates with a poor
prognosis. APMIS Acta Pathol Microbiol Immunol Scand. 2013 Nov;121(11):1047–53.
238. Liu Y, Zhao
L, Ma W, Cao X, Chen H, Feng D, et al. The Blockage of KCa3.1 Channel Inhibited
Proliferation, Migration and Promoted Apoptosis of Human Hepatocellular
Carcinoma Cells. J Cancer. 2015;6(7):643–51.
239. D’Alessandro
G, Catalano M, Sciaccaluga M, Chece G, Cipriani R, Rosito M, et al. KCa3.1
channels are involved in the infiltrative behavior of glioblastoma in vivo.
Cell Death Dis. 2013 Aug;4(8):e773–e773.
240. Chigurupati
S, Venkataraman R, Barrera D, Naganathan A, Madan M, Paul L, et al. Receptor
channel TRPC6 is a key mediator of Notch-driven glioblastoma growth and
invasiveness. Cancer Res. 2010 Jan;70(1):418–27.
241. Bomben VC,
Sontheimer HW. Inhibition of transient receptor potential canonical channels
impairs cytokinesis in human malignant gliomas. Cell Prolif. 2008
Feb;41(1):98–121.
242. Lee YS,
Sayeed MM, Wurster RD. Inhibition of cell growth and intracellular Ca2+
mobilization in human brain tumor cells by Ca2+ channel antagonists. Mol Chem
Neuropathol. 1994 Jun;22(2):81–95.
243. Venturini E,
Leanza L, Azzolini M, Kadow S, Mattarei A, Weller M, et al. Targeting the
Potassium Channel Kv1.3 Kills Glioblastoma Cells. Neurosignals.
2017;25(1):26–38.
244. Huang L, Li
B, Tang S, Guo H, Li W, Huang X, et al. Mitochondrial KATP Channels Control
Glioma Radioresistance by Regulating ROS-Induced ERK Activation. Mol Neurobiol.
2015 Aug;52(1):626–37.
245. Visa A,
Sallán MC, Maiques O, Alza L, Talavera E, López-Ortega R, et al. T-Type Cav3. 1
channels mediate progression and chemotherapeutic resistance in glioblastoma.
Cancer Res. 2019;79(8):1857–68.
246. Liu S, Ba Y,
Li C, Xu G. Inactivation of CACNA1H induces cell apoptosis by initiating
endoplasmic reticulum stress in glioma. Transl Neurosci. 2023;14(1):20220285.
247. Valerie NC,
Dziegielewska B, Hosing AS, Augustin E, Gray LS, Brautigan DL, et al.
Inhibition of T-type calcium channels disrupts Akt signaling and promotes
apoptosis in glioblastoma cells. Biochem Pharmacol. 2013;85(7):888–97.
248. Mertens-Walker
I, Bolitho C, Baxter RC, Marsh DJ. Gonadotropin-induced ovarian cancer cell
migration and proliferation require extracellular signal-regulated kinase 1/2
activation regulated by calcium and protein kinase Cδ. Endocr Relat Cancer.
2010;17(2):335.
249. Bomben VC,
Turner KL, Barclay TC, Sontheimer H. Transient receptor potential canonical
channels are essential for chemotactic migration of human malignant gliomas. J
Cell Physiol. 2011;226(7):1879–88.
250. Cuddapah VA,
Turner KL, Sontheimer H. Calcium entry via TRPC1 channels activates chloride
currents in human glioma cells. Cell Calcium. 2013;53(3):187–94.
251. Richter JM,
Schaefer M, Hill K. Riluzole activates TRPC5 channels independently of PLC
activity. Br J Pharmacol. 2014;171(1):158–70.
252. Zeng J, Wu
Y, Zhuang S, Qin L, Hua S, Mungur R, et al. Identification of the role of TRPM8
in glioblastoma and its effect on proliferation, apoptosis and invasion of the
U251 human glioblastoma cell line. Oncol Rep. 2019;42(4):1517–26.
253. Caceres AI,
Liu B, Jabba SV, Achanta S, Morris JB, Jordt S. Transient receptor potential
cation channel subfamily M member 8 channels mediate the anti‐inflammatory
effects of eucalyptol. Br J Pharmacol. 2017;174(9):867–79.
254. Wang S, Li
X, Hu Y, Wang L, Lv G, Feng Y, et al. Discovery of N-alkyl-N-benzyl thiazoles
as novel TRPC antagonists for the treatment of glioblastoma multiforme. Eur J
Med Chem. 2024;265:116066.
255. Wan J, Guo
AA, King P, Guo S, Saafir T, Jiang Y, et al. TRPM7 induces tumorigenesis and
stemness through notch activation in glioma. Front Pharmacol. 2020;11:590723.
256. Yin H, Cheng
H, Li P, Yang Z. TRPC6 interacted with KCa1. 1 channels to regulate the
proliferation and apoptosis of glioma cells. Arch Biochem Biophys.
2022;725:109268.
257. Freeman S,
Elzamzamy O, Geldenhuys W, Hazlehurst L. CLIC1 is an Attractive Candidate for
the Initial Binding Target of the Novel Peptide MTI‐101. FASEB J.
2020;34(S1):1–1.
258. Xiong D,
Heyman NS, Airey J, Zhang M, Singer CA, Rawat S, et al. Cardiac-specific,
inducible ClC-3 gene deletion eliminates native volume-sensitive chloride
channels and produces myocardial hypertrophy in adult mice. J Mol Cell Cardiol.
2010;48(1):211–9.
259. Morelli MB,
Nabissi M, Amantini C, Maggi F, Ricci-Vitiani L, Pallini R, et al. TRPML2
Mucolipin Channels Drive the Response of Glioma Stem Cells to Temozolomide and
Affect the Overall Survival in Glioblastoma Patients. Int J Mol Sci.
2022;23(23):15356.
260. Santoni G,
Maggi F, Amantini C, Arcella A, Marinelli O, Nabissi M, et al. Coexpression of
TRPML1 and TRPML2 Mucolipin Channels Affects the Survival of Glioblastoma
Patients. Int J Mol Sci. 2022;23(14):7741.
261. Mori A,
Lehmann S, O’Kelly J, Kumagai T, Desmond JC, Pervan M, et al. Capsaicin, a
component of red peppers, inhibits the growth of androgen-independent, p53
mutant prostate cancer cells. Cancer Res. 2006;66(6):3222–9.
262. Prevarskaya
N, Zhang L, Barritt G. TRP channels in cancer. Biochim Biophys Acta BBA-Mol
Basis Dis. 2007;1772(8):937–46.
263. Huang T, Xu
T, Wang Y, Zhou Y, Yu D, Wang Z, et al. Cannabidiol inhibits human glioma by
induction of lethal mitophagy through activating TRPV4. Autophagy.
2021;17(11):3592–606.
264. Yang W, Wu P
fei, Ma J xing, Liao M jun, Xu L shan, Yi L. TRPV4 activates the Cdc42/N-wasp
pathway to promote glioblastoma invasion by altering cellular protrusions. Sci
Rep. 2020;10(1):14151.
265. Thorneloe
KS, Cheung M, Bao W, Alsaid H, Lenhard S, Jian MY, et al. An orally active
TRPV4 channel blocker prevents and resolves pulmonary edema induced by heart
failure. Sci Transl Med. 2012;4(159):159ra148-159ra148.
266. Venturini E,
Leanza L, Azzolini M, Kadow S, Mattarei A, Weller M, et al. Targeting the
potassium channel Kv1. 3 kills glioblastoma cells. Neurosignals.
2018;25(1):26–38.
267. Grimaldi A,
D’Alessandro G, Di Castro MA, Lauro C, Singh V, Pagani F, et al. Kv1. 3
activity perturbs the homeostatic properties of astrocytes in glioma. Sci Rep.
2018;8(1):7654.
268. Leonard RJ,
Garcia ML, Slaughter RS, Reuben JP. Selective blockers of voltage-gated K+
channels depolarize human T lymphocytes: mechanism of the antiproliferative
effect of charybdotoxin. Proc Natl Acad Sci. 1992;89(21):10094–8.
269. Staudacher
I, Jehle J, Staudacher K, Pledl HW, Lemke D, Schweizer PA, et al. HERG K+
channel-dependent apoptosis and cell cycle arrest in human glioblastoma cells.
PloS One. 2014;9(2):e88164.
270. Shugg T,
Dave N, Amarh E, Assiri AA, Pollok KE, Overholser BR. Letrozole targets the
human ether‐a‐go‐go–related gene potassium current in glioblastoma. Basic Clin
Pharmacol Toxicol. 2021;128(3):357–65.
271. Cherubini A,
Taddei G, Crociani O, Paglierani M, Buccoliero A, Fontana L, et al. HERG
potassium channels are more frequently expressed in human endometrial cancer as
compared to non-cancerous endometrium. Br J Cancer. 2000;83(12):1722–9.
272. Caramia M,
Sforna L, Franciolini F, Catacuzzeno L. The volume-regulated anion channel in
glioblastoma. Cancers. 2019;11(3):307.
273. Lv J, Liang
Y, Zhang S, Lan Q, Xu Z, Wu X, et al. DCPIB, an inhibitor of volume-regulated
anion channels, distinctly modulates K2P channels. ACS Chem Neurosci.
2019;10(6):2786–93.
274. Voss FK,
Ullrich F, Münch J, Lazarow K, Lutter D, Mah N, et al. Identification of LRRC8
heteromers as an essential component of the volume-regulated anion channel
VRAC. Science. 2014;344(6184):634–8.
275. Baker EM,
Thompson CH, Hawkins NA, Wagnon JL, Wengert ER, Patel MK, et al. The novel
sodium channel modulator GS‐458967 (GS 967) is an effective treatment in a
mouse model of SCN 8A encephalopathy. Epilepsia. 2018;59(6):1166–76.
276. Xing D, Wang
J, Ou S, Wang Y, Qiu B, Ding D, et al. Expression of neonatal Nav1. 5 in human
brain astrocytoma and its effect on proliferation, invasion and apoptosis of
astrocytoma cells. Oncol Rep. 2014;31(6):2692–700.
277. O’Hara T,
Rudy Y. Quantitative comparison of cardiac ventricular myocyte
electrophysiology and response to drugs in human and nonhuman species. Am J
Physiol-Heart Circ Physiol. 2012;302(5):H1023–30.
278. Ramaswamy P,
Dalavaikodihalli Nanjaiah N, Prasad C, Goswami K. Transcriptional modulation of
calcium‐permeable AMPA receptor subunits in glioblastoma by MEK–ERK1/2
inhibitors and their role in invasion. Cell Biol Int. 2020;44(3):830–7.
279. Arai A,
Kessler M. Pharmacology of ampakine modulators: from AMPA receptors to synapses
and behavior. Curr Drug Targets. 2007;8(5):583–602.
280. Partin KM,
Fleck MW, Mayer ML. AMPA receptor flip/flop mutants affecting deactivation,
desensitization, and modulation by cyclothiazide, aniracetam, and thiocyanate.
J Neurosci. 1996;16(21):6634–47.
281. Kim HJ, Kim
JY, Jung CW, Lee YS, An JY, Kim EH, et al. ANO1 regulates the maintenance of
stemness in glioblastoma stem cells by stabilizing EGFRvIII. Oncogene.
2021;40(8):1490–502.
282. Xuan Z bo,
Wang Y ji, Xie J. ANO6 promotes cell proliferation and invasion in glioma
through regulating the ERK signaling pathway. OncoTargets Ther. 2019;6721–31.
283. Britschgi A,
Bill A, Brinkhaus H, Rothwell C, Clay I, Duss S, et al. Calcium-activated
chloride channel ANO1 promotes breast cancer progression by activating EGFR and
CAMK signaling. Proc Natl Acad Sci. 2013;110(11):E1026–34.
284. Sheng Y, Wu
B, Leng T, Zhu L, Xiong Z. Acid-sensing ion channel 1 (ASIC1) mediates weak
acid-induced migration of human malignant glioma cells. Am J Cancer Res.
2021;11(3):997.
285. King P, Wan
J, Guo AA, Guo S, Jiang Y, Liu M. Regulation of gliomagenesis and stemness
through acid sensor ASIC1a. Int J Oncol. 2021;59(4):1–15.
286. Leng T, Si
H, Li J, Yang T, Zhu M, Wang B, et al. Amiloride Analogs as ASIC 1a Inhibitors.
CNS Neurosci Ther. 2016;22(6):468–76.
287. Kapoor N,
Lee W, Clark E, Bartoszewski R, McNicholas CM, Latham CB, et al. Interaction of
ASIC1 and ENaC subunits in human glioma cells and rat astrocytes. Am J
Physiol-Cell Physiol. 2011;300(6):C1246–59.
288. Chen WC, Kuo
TH, Tzeng YS, Tsai YC. Baicalin induces apoptosis in SW620 human colorectal
carcinoma cells in vitro and suppresses tumor growth in vivo. Molecules.
2012;17(4):3844–57.
289. Huang L, Li
B, Li W, Guo H, Zou F. ATP-sensitive potassium channels control glioma cells
proliferation by regulating ERK activity. Carcinogenesis. 2009;30(5):737–44.
290. Hattersley
AT, Ashcroft FM. Activating mutations in Kir6. 2 and neonatal diabetes: new
clinical syndromes, new scientific insights, and new therapy. Diabetes.
2005;54(9):2503–13.
291. Rafiq M,
Flanagan SE, Patch AM, Shields BM, Ellard S, Hattersley AT, et al. Effective
treatment with oral sulfonylureas in patients with diabetes due to sulfonylurea
receptor 1 (SUR1) mutations. Diabetes Care. 2008;31(2):204–9.
292. D’Alessandro
G, Limatola C, Catalano M. Functional roles of the Ca2+-activated K+ channel,
KCa3. 1, in brain tumors. Curr Neuropharmacol. 2018;16(5):636–43.
293. Gribkoff VK,
Starrett Jr JE, Dworetzky SI. The Pharmacology and Molecular Biology of
Large-Conductance Calcium= Activated (BK) Potassium Channels. Adv Pharmacol.
1996;37:319–48.
294. Zhou Y,
Lingle CJ. Paxilline inhibits BK channels by an almost exclusively
closed-channel block mechanism. J Gen Physiol. 2014;144(5):415–40.
295. Ataga KI,
Smith WR, De Castro LM, Swerdlow P, Saunthararajah Y, Castro O, et al. Efficacy
and safety of the Gardos channel blocker, senicapoc (ICA-17043), in patients
with sickle cell anemia. Blood J Am Soc Hematol. 2008;111(8):3991–7.
296. Zhou W, Liu
X, van Wijnbergen JWM, Yuan L, Liu Y, Zhang C, et al. Identification of PIEZO1
as a potential prognostic marker in gliomas. Sci Rep. 2020;10(1):16121.
297. Chen X,
Wanggou S, Bodalia A, Zhu M, Dong W, Fan JJ, et al. A feedforward mechanism
mediated by mechanosensitive ion channel PIEZO1 and tissue mechanics promotes
glioma aggression. Neuron. 2018;100(4):799–815.
298. Bae C, Sachs
F, Gottlieb PA. The mechanosensitive ion channel Piezo1 is inhibited by the
peptide GsMTx4. Biochemistry. 2011;50(29):6295–300.
299. Kurata T,
Rajendran V, Fan S, Ohta T, Numata M, Fushida S. NHE5 regulates growth factor
signaling, integrin trafficking, and degradation in glioma cells. Clin Exp
Metastasis. 2019;36:527–38.
300. Ko M, Makena
MR, Schiapparelli P, Suarez-Meade P, Mekile AX, Lal B, et al. The endosomal pH
regulator NHE9 is a driver of stemness in glioblastoma. PNAS Nexus.
2022;1(1):pgac013.
301. Pall AE,
Juratli L, Guntur D, Bandyopadhyay K, Kondapalli KC. A gain of function
paradox: Targeted therapy for glioblastoma associated with abnormal NHE9
expression. J Cell Mol Med. 2019;23(11):7859–72.
302. Luo L, Wang
J, Ding D, Hasan MN, Yang SS, Lin SH, et al. Role of NKCC1 activity in glioma
K+ homeostasis and cell growth: New insights with the bumetanide-derivative
STS66. Front Physiol. 2020;11:911.
303. Blaesse P,
Airaksinen MS, Rivera C, Kaila K. Cation-chloride cotransporters and neuronal
function. Neuron. 2009;61(6):820–38.
304. Nieto FR,
Cobos EJ, Tejada MÁ, Sánchez-Fernández C, González-Cano R, Cendán CM.
Tetrodotoxin (TTX) as a therapeutic agent for pain. Mar Drugs. 2012
Feb;10(2):281–305.
305. Challapalli
V, Tremont-Lukats IW, McNicol ED, Lau J, Carr DB. Systemic administration of
local anesthetic agents to relieve neuropathic pain. Cochrane Database Syst
Rev. 2019 Oct;2019(10).
306. Brinkrolf P,
Hahnenkamp K. Systemic lidocaine in surgical procedures: effects beyond sodium
channel blockade. Curr Opin Anaesthesiol. 2014 Aug;27(4):420–5.
307. McCann G.
Pharmacological treatment of significant cardiac arrhythmias. Br J Sports Med.
2000 Oct;34(5):401–2.
308. Fairhurst C,
Martin F, Watt I, Doran T, Bland M, Brackenbury WJ. Sodium channel-inhibiting
drugs and cancer survival: protocol for a cohort study using the CPRD primary
care database. BMJ Open. 2016 Sep;6(9):e011661–e011661.
309. Doty P, Rudd
GD, Stoehr T, Thomas D. Lacosamide. Neurother J Am Soc Exp Neurother. 2007
Jan;4(1):145–8.
310. Mantegazza
M, Curia G, Biagini G, Ragsdale DS, Avoli M. Voltage-gated sodium channels as
therapeutic targets in epilepsy and other neurological disorders. Lancet
Neurol. 2010 Apr;9(4):413–24.
311. Black JA,
Waxman SG. Phenytoin protects central axons in experimental autoimmune
encephalomyelitis. J Neurol Sci. 2008 Nov;274(1–2):57–63.
312. Perucca E. A
pharmacological and clinical review on topiramate, a new antiepileptic drug.
Pharmacol Res. 1997 Apr;35(4):241–56.
313. Silke B,
Goldhammer E, Sharma SK, Verma SP, Taylor SH. An exercise hemodynamic
comparison of verapamil, diltiazem, and amlodipine in coronary artery disease.
Cardiovasc Drugs Ther. 1990 Apr;4(2):457–63.
314. Rubaiy HN.
The therapeutic agents that target ATP-sensitive potassium channels. Acta Pharm
Zagreb Croat. 2016 Mar;66(1):23–34.
315. Sanguinetti
MC, Bennett PB. Antiarrhythmic drug target choices and screening. Circ Res.
2003 Sep;93(6):491–9.
316. Ravens U,
Poulet C, Wettwer E, Knaut M. Atrial selectivity of antiarrhythmic drugs. J
Physiol. 2013 Sep;591(17):4087–97.
317. Michiels CF,
Van Hove CE, Martinet W, De Meyer GRY, Fransen P. L-type Ca2+ channel blockers
inhibit the window contraction of mouse aorta segments with high affinity. Eur
J Pharmacol. 2014 Sep;738:170–8.
318. Lukasova M,
Malaval C, Gille A, Kero J, Offermanns S. Nicotinic acid inhibits progression
of atherosclerosis in mice through its receptor GPR109A expressed by immune
cells. J Clin Invest. 2011 Mar;121(3):1163–73.
319. Ashcroft SJ,
Ashcroft FM. Properties and functions of ATP-sensitive K-channels. Cell Signal.
1990;2(3):197–214.
320. Jang SH,
Choi SY, Ryu PD, Lee SY. Anti-proliferative effect of Kv1.3 blockers in A549
human lung adenocarcinoma in vitro and in vivo. Eur J Pharmacol. 2011
Jan;651(1–3):26–32.
321. Borrelli F,
Pagano E, Romano B, Panzera S, Maiello F, Coppola D, et al. Colon
carcinogenesis is inhibited by the TRPM8 antagonist cannabigerol, a
Cannabis-derived non-psychotropic cannabinoid. Carcinogenesis. 2014
Dec;35(12):2787–97.
322. Huang MH,
Huang YM, Wu SN. The Inhibition by Oxaliplatin, a Platinum-Based
Anti-Neoplastic Agent, of the Activity of Intermediate-Conductance Ca2+-Activated
K+ Channels in Human Glioma Cells. Cell Physiol Biochem Int J Exp
Cell Physiol Biochem Pharmacol. 2015;37(4):1390–406.
323. Djamgoz MBA,
Onkal R. Persistent current blockers of voltage-gated sodium channels: a
clinical opportunity for controlling metastatic disease. Recent Patents
Anticancer Drug Discov. 2013 Jan;8(1):66–84.
324. Brackenbury
WJ, Chioni AM, Diss JKJ, Djamgoz MBA. The neonatal splice variant of Nav1.5
potentiates in vitro invasive behaviour of MDA-MB-231 human breast cancer
cells. Breast Cancer Res Treat. 2007 Jan;101(2):149–60.
325. Yavuz M,
Şahin B, Baykal AT, Demircan T. Hydroquinidine displays a significant
anti-carcinogenic activity in breast and ovarian cancer cells via inhibiting
cell-cycle and stimulating apoptosis. Turk J Biol. 2023;47(1):44–60.
326. Dao Trong P,
Jungwirth G, Unterberg A, Herold-Mende C, Warta R. The Antiepileptic Drug
Oxcarbazepine Inhibits the Growth of Patient-Derived Isocitrate Dehydrogenase
Mutant Glioma Stem-like Cells. Cells. 2023;12(8):1200.
327. Dong W,
Fekete A, Chen X, Liu H, Beilhartz GL, Chen X, et al. A designer peptide
against the EAG2–Kvβ2 potassium channel targets the interaction of cancer cells
and neurons to treat glioblastoma. Nat Cancer. 2023;4(10):1418–36.
328. Mlayah-Bellalouna
S, Aissaoui-Zid D, Chantome A, Jebali J, Souid S, Ayedi E, et al. Insights into
the mechanisms governing P01 scorpion toxin effect against U87 glioblastoma
cells oncogenesis. Front Pharmacol. 2023;14:1203247.
329. Dinevska M,
Gazibegovic N, Morokoff AP, Kaye AH, Drummond KJ, Mantamadiotis T, et al.
Inhibition of radiation and temozolomide-induced glioblastoma invadopodia
activity using ion channel drugs. Cancers. 2020;12(10):2888.
330. Luo L, Wang
J, Ding D, Hasan MN, Yang SS, Lin SH, et al. Role of NKCC1 activity in glioma
K+ homeostasis and cell growth: New insights with the bumetanide-derivative
STS66. Front Physiol. 2020;11:911.
331. Huang T, Xu
T, Wang Y, Zhou Y, Yu D, Wang Z, et al. Cannabidiol inhibits human glioma by
induction of lethal mitophagy through activating TRPV4. Autophagy.
2021;17(11):3592–606.